Our Environment Class 10 Biology Notes: Chapter 13 Our Environment is an important topic in Class 10 Science that helps students understand how living organisms interact with their surroundings. In Chapter 13 Our Environment Class 10 Biology notes, students learn about ecosystems, food chains, food webs, trophic levels, and the flow of energy in nature. These concepts explain how plants, animals, microorganisms, air, water, and soil are connected with each other and maintain balance in the environment.
These our environment class 10th notes are designed to help students clearly understand the structure and function of ecosystems. The chapter also explains how energy moves from producers to consumers and then to decomposers. Understanding these topics is very important because it helps students know how natural systems work and why environmental balance is necessary for life on Earth.
In these our environment class 10 biology notes, students will also explore topics like biological magnification, waste management, and the role of human activities in environmental changes. These ideas are commonly asked in exams, so practicing our environment class 10 important questions and reviewing our environment class 10 questions and answers can help students prepare better for board exams.
Overall, this chapter builds a basic understanding of ecology and environmental science. It also encourages students to think about protecting nature and using resources responsibly, which is very important for a sustainable future. Sometimes students think ecosystem topics are complex, but with proper notes it becomes much easier to understand.
What Is Our Environment?
The environment encompasses everything that surrounds an organism and influences its life the complete physical and biological world in which living beings exist. Understanding the environment is fundamental to appreciating how life on Earth is sustained, balanced, and sometimes threatened.
Our environment comprises three key components:
- Living organisms – plants, animals, human beings, and microorganisms
- Physical surroundings – land, water bodies, and air
- Meteorological (climatic) factors – sunlight, temperature, rainfall, and humidity
CBSE Class 10 Biology Notes - Our Environment Revision Notes PDF
Fill the form to download this PDF
Ecosystem
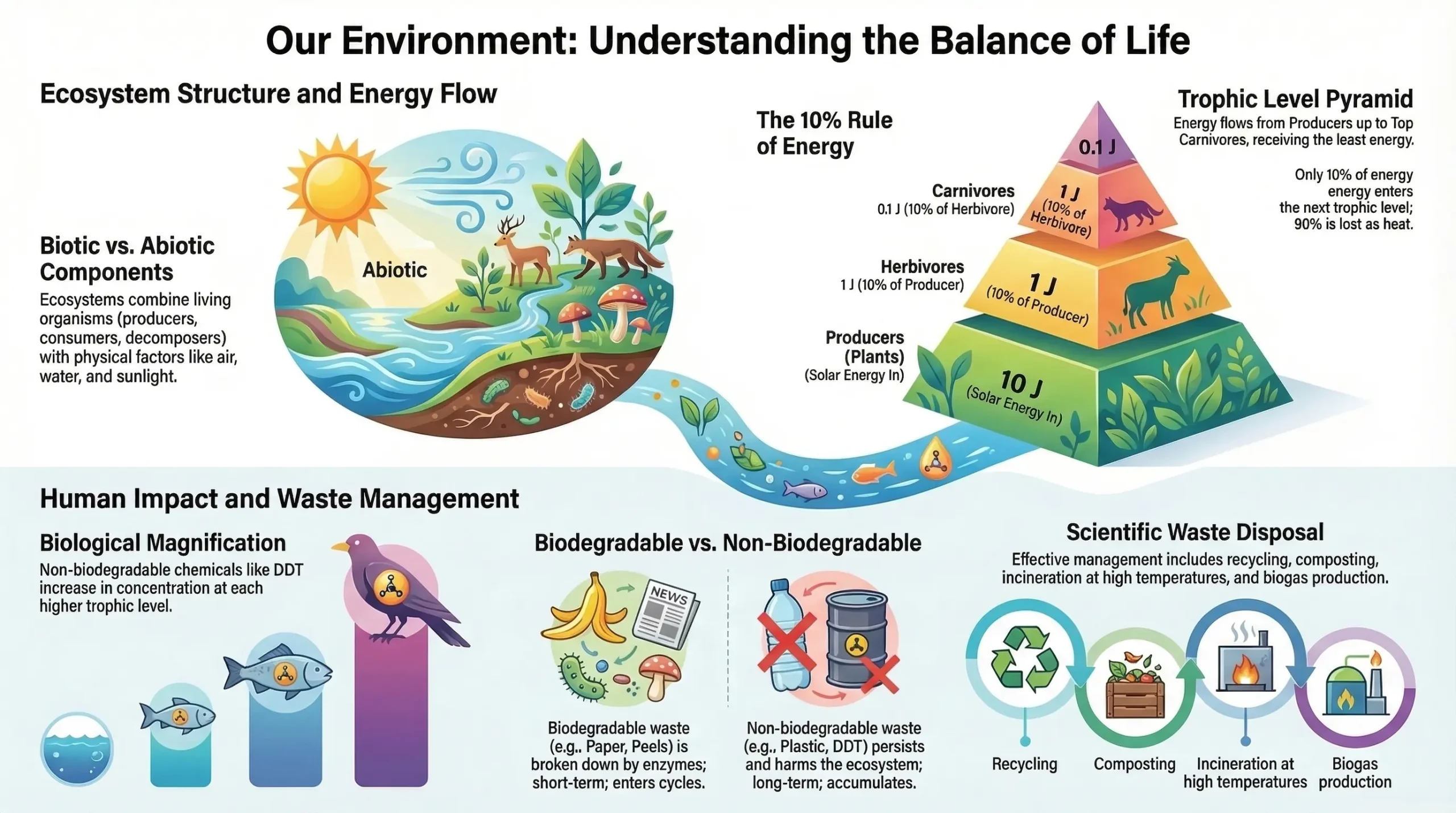
An ecosystem is a structural and functional unit of the biosphere. It consists of living organisms and their physical environment, which interact with each other and maintain a balance in nature. In every ecosystem, energy and matter are continuously exchanged between living (biotic) and non-living (abiotic) components.
Types of Ecosystems
Ecosystems are broadly classified as:
Natural Ecosystems
- Aquatic – Fresh Water (Lentic: ponds, lakes, Lotic: rivers, streams, Wet Lands: marshes, swamps) and Marine (Coastal Areas, Oceans)
- Terrestrial – Grassland, Desert, Forest
Artificial Ecosystems
- Aquarium, Cropland
Components of an Ecosystem
Every ecosystem contains two fundamental types of components:
2.1 Abiotic (Non-Living) Components
Abiotic components are the non-living physico-chemical factors that affect the distribution, structure, behaviour, and interrelationships of organisms. They include:
- Inorganic substances: Carbon, nitrogen, oxygen, calcium, phosphorus, water, carbon dioxide found dissolved in water, in soil, or as free gases in the air.
- Organic substances: Carbohydrates, proteins, lipids, nucleic acids present in living organisms and dead organic matter. Decomposers break these down into inorganic substances for recycling.
- Climatic (Edaphic) factors: Light, temperature, humidity, wind, rainfall, soil pH, topography, minerals.
Biotic (Living) Components
The living organisms of an ecosystem form the biotic component. Based on their mode of nutrition, they are classified into:
(i) Producers (Autotrophs)
All green plants and blue-green algae are producers. They use solar energy with the help of photosynthetic pigments (e.g., chlorophyll) to synthesize glucose from carbon dioxide and water a process called photosynthesis:
6CO₂ + 6H₂O →(Sunlight/Chlorophyll)→ C₆H₁₂O₆ + 6O₂
Because they prepare their own organic food using sunlight, they are called photoautotrophs (or simply autotrophs). From glucose, plants synthesize complex compounds like starch, proteins, and lipids.
(ii) Consumers (Heterotrophs)
Animals that cannot synthesize their own food. They depend on producers or other organisms for energy. Classified as:
| Consumer Type | Also Called | Feeds On | Examples (Terrestrial) | Examples (Aquatic) |
|---|---|---|---|---|
| Primary (1st order) | Herbivores | Plants | Cattle, deer, rabbit, grasshopper | Snails, tadpoles, tortoises |
| Secondary (2nd order) | Carnivores | Herbivores | Cats, dogs, foxes | Water bugs, frogs, small fish |
| Tertiary (3rd order) | Larger Carnivores | Secondary consumers | Wolves, snakes | Large fish, water birds |
| Quaternary (4th order) | Top Carnivores | Tertiary consumers | Tigers, lions | Dolphins, whales |
Omnivores: Animals that consume both plants and animals (e.g., crow, bear, dog, humans) also called double consumers.
Parasites: Organisms that live on/inside another organism to obtain food (e.g., Escherichia coli, tapeworm, liverfluke).
(iii) Decomposers (Reducers / Saprotrophs)
Decomposers obtain food from organic materials of dead producers and consumers and their waste products. They degrade dead remains into simple inorganic substances, releasing them back into the environment (biogeochemical cycles) for reuse by producers.
Examples: Bacteria and Fungi
Importance of Decomposers:
- Act as cleansing agents by decomposing dead organisms.
- Return nutrient elements to soil, air, and water for reuse by producers.
- Maintain soil fertility.
- Facilitate recycling of materials in the biosphere — keeping life going as an unending chain.
Food Chain
A food chain is the sequential interlinking of organisms involving the transfer of food energy from producers, through a series of organisms with repeated eating and being eaten.
Examples of Food Chains
Grassland/Forest:
Grass → Deer → Lion (Producer) (Herbivore) (Carnivore)
Aquatic (Pond/Lake):
Algae → Zooplankton → Small Fish → Big Fish (Producer) (Herbivore) (Small Carnivore) (Big Carnivore)
Algae → Protozoan → Fish → Squil → Seal (Phytoplankton) (Zooplankton) (Carnivore I) (Carnivore II) (Top Carnivore)
Note on Planktons:
- Phytoplanktons – microscopic aquatic plants (e.g., algae); act as producers
- Zooplanktons – microscopic aquatic animals (e.g., protozoans); act as herbivores feeding on phytoplanktons
3.2 Types of Food Chains
Grazing Food Chain – begins with living green plants as producers:
| Ecosystem | Producers | Herbivores | Primary Carnivores | Secondary Carnivores | Tertiary Carnivores |
|---|---|---|---|---|---|
| Grassland | Grass | Insects, Rats, Rabbits | Frog, Snakes, Foxes | Snakes, Predatory birds, Wolves | Predatory birds, Lion |
| Pond | Phytoplanktons | Zooplanktons | Small fish | Large fish | Predatory birds |
Detritus Food Chain – begins with dead organic matter (detritus):
| Detritus | Detritivores | Detritivore Consumers | Small Carnivores | Large Carnivores |
|---|---|---|---|---|
| Fallen leaves and dead bodies | Fungi, Bacteria, Protozoans | Insect Larvae, Crustaceans, Molluscs | Minnows, Small fish | Large fish, Fish-eating birds |
3.3 Characteristics of a Food Chain
- Involves nutritive interaction between biotic components of an ecosystem.
- Always straight and proceeds in a progressive linear direction.
- Energy flows unidirectionally from sun → producers → consumers.
- Typically 3–4 trophic levels; maximum 5 trophic levels in rare cases.
- Omnivores occupy different trophic positions in different food chains.
- At each transfer, 80–90% of energy is lost as heat; only 10% is transferred to the next trophic level (Second Law of Thermodynamics).
Trophic Levels
The distinct sequential steps in a food chain where energy transfer occurs are called trophic levels.
| Trophic Level | Organisms | Alternate Name |
|---|---|---|
| 1st | Green plants (Producers) | Producer level |
| 2nd | Plant-eaters (Herbivores) | Primary consumers |
| 3rd | Flesh-eaters (Carnivores) | Secondary consumers |
| 4th | Larger carnivores | Tertiary consumers |
| 5th | Top carnivores | Quaternary consumers |
Food Web
A food web is a network of interconnected food chains at various trophic levels, forming complex feeding relationships among different organisms in a biotic community.
In nature, organisms are generally eaten by more than one type of organism, creating an interlocking pattern rather than a straight line. In a food web, any given species may operate at more than one trophic level simultaneously.
Characteristics of a Food Web
- Food webs are never straight they are formed by interlinking of multiple food chains.
- Provide alternative food pathways if one species of producer is destroyed, herbivores can feed on other producers.
- Greater alternatives in a food web make the ecosystem more stable.
- Help check overpopulation of highly productive species.
- Contribute to ecosystem development.
Ecological Pyramid
An ecological pyramid is a graphical representation of a specific parameter (number, biomass, or energy) at each trophic level of a food chain. The concept was introduced by Charles Elton in 1927.
Pyramid of Number
Shows the number of individuals at each trophic level.
- Upright: Grassland and pond ecosystems
- Intermediate: Forest ecosystem
- Inverted (Spindle-shaped): Parasitic food chains (e.g., Tree → Birds → Parasites — number increases at higher trophic levels)
Biotic potential = The number of members of a species thriving under favorable conditions. Rapid increase in members of a species is called a population explosion.
Pyramid of Biomass
Shows total dry weight (biomass) of organisms at each trophic level.
- Upright: Grassland and forest ecosystems
- Inverted: Pond ecosystem (aquatic biomass is concentrated in small producers)
Example values (upright):
- Producers: 1000 kg | Herbivores: 100 kg | Carnivores: 10 kg | Top Carnivores: 1 kg
Pyramid of Energy
Shows the energy content or productivity at different trophic levels.
- Always upright in all ecosystems — without exception.
Example values:
- Vegetation: 1000 J | Mice: 100 J | Snakes: 10 J | Hawk: 1 J
Flow of Energy in an Ecosystem
Energy flow in ecosystems obeys the Laws of Thermodynamics energy is not created but only converted from one form to another.
- About 1% of solar energy reaching leaves is used in photosynthesis.
- Energy flows unidirectionally from sun → producers → consumers.
- At every trophic level, energy is used for metabolic activities (respiration, growth) and for decomposers.
- The energy lost as heat cannot be reused by plants.
The 10% Law
Proposed by Raymond Lindeman in 1942: Only 10% of the total energy entering a particular trophic level is transferred to the next trophic level.
Example with 1000 J of solar energy:
- Plants (Producers) trap: 10 J (1% of 1000 J); 990 J lost to environment
- Herbivores receive: 1 J (10% of 10 J); 9 J lost to environment
- Carnivores receive: 0.1 J (10% of 1 J); 0.9 J lost to environment
Flow of material is cyclic; flow of energy is unidirectional.
Biological Magnification (Biomagnification)
Biological magnification is the progressive increase in concentration of harmful chemical substances in the bodies of living organisms at each successive trophic level of a food chain.
How It Happens
Non-biodegradable toxic chemicals such as DDT (pesticide) and heavy metals (mercury, arsenic, cadmium) enter organisms through food chains and accumulate at each trophic level.
Example – DDT Concentration in an aquatic food chain:
| Organism | DDT Concentration |
|---|---|
| Water | 0.02 ppm |
| Phytoplanktons/Zooplanktons | 5 ppm |
| Fish | 240 ppm |
| Fish-eating Birds | 1600 ppm |
Since humans occupy the highest trophic level, they accumulate the maximum concentration of harmful chemicals.
Health Impacts
- Mercury accumulation → Minamata disease (mercury poisoning)
- Fluorine accumulation → Knee-knock syndrome
- Pesticides → Carcinogenic effects, hormonal disruption
Human Activities Affecting the Environment
Several human activities disturb natural environmental balance:
- Deforestation – for fuel, furniture, agriculture, and urbanization
- Mining – extraction of minerals, coal, and petroleum
- Transportation – large-scale use of vehicles
- Power generation – thermal, hydroelectric, and nuclear power plants
- Infrastructure development – roads, railway tracks
- Industrial chemical production – harmful products and effluents
- Extensive pesticide use
- Use of Chlorofluorocarbons (CFCs)
Major resulting environmental problems: Ozone layer depletion, Waste disposal, Acid rain, Global warming.
Solid Wastes
Solid wastes originate from residences, industries, cattle sheds, and agricultural fields. They include fruit/vegetable peelings, kitchen waste, ash, paper, glass, plastics, leather, rubber, metals, and human excreta.
Types of Solid Wastes
Biodegradable Wastes
Substances that can be broken down by microorganisms (bacteria, fungi) using specific enzymes into simpler, harmless substances. They re-enter biogeochemical cycles.
Examples: Urine, faecal matter, sewage, paper, vegetable peels, agricultural residues, wood, cloth.
Harmful effects (when accumulated):
- Production of foul smell → health hazard
- Fly breeding → spread of diarrhea, typhoid, tuberculosis, cholera, conjunctivitis
- Drain blockage → mosquito breeding → malaria and dengue
- Industrial waste dumping → reduced soil fertility and crop yield
Non-Biodegradable Wastes
Substances that cannot be broken down by biological agents; only physical processes (heat, pressure) can affect them. They persist in the environment for long periods.
Examples: Heavy metals (mercury, lead, arsenic), radioactive wastes (uranium, plutonium), insecticides and pesticides (DDT, BHC).
Harmful effects:
- Enter food chains → biomagnification → toxic effects in humans and animals
- Reduce soil fertility → crop yield decreases → soil becomes acidic or alkaline
Comparison: Biodegradable vs Non-Biodegradable Wastes
| Feature | Biodegradable Waste | Non-Biodegradable Waste |
|---|---|---|
| Degradation | By biological enzymes (bacteria, fungi) | Only by physical processes (heat, pressure) |
| Biogeochemical cycles | Can enter cycles | Cannot enter cycles |
| Pollution trigger | Only when accumulated in excess | Always act as pollutants |
| Persistence | Short-lived in environment | Persist for a long time |
| Management | Treat before disposal | Recycle or reuse |
| Examples | Vegetable peels, sewage, paper | DDT, mercury, uranium, BHC |
Managing Garbage
Garbage = household waste including leftover food, waste paper, plastic bags. Improper disposal pollutes soil, water, and air.
Modes of Waste Disposal
- Landfills: Burying solid wastes in low-lying areas to level uneven land surfaces.
- Recycling: Paper → paper mills; plastic → plastic processing factories; metals → metal industries.
- Composting: Converting kitchen waste, vegetable peels, and fallen leaves into manure.
- Incineration: Burning waste at high temperature (>1000°C) in an incinerator → produces CO₂, water vapour, and ash. Used for household, chemical, and hospital waste.
- Biogas production: Biodegradable waste → biogas plants → biogas (fuel) + manure (fertilizer).
Greenhouse Effect and Global Warming
A greenhouse is a glass enclosure where temperature inside remains higher than outside. Greenhouse gases in the atmosphere behave similarly they allow sunlight in but trap the heat radiating from Earth's surface.
Greenhouse Gases
- Carbon dioxide (CO₂)
- Chlorofluorocarbons (CFCs)
- Methane (CH₄)
- Nitrous oxides (N₂O₅)
Global warming = the rise in Earth's average temperature due to increased concentration of greenhouse gases trapping long-wave radiation.
Causes of Global Warming
| Greenhouse Gas | Main Human Sources |
|---|---|
| CO₂ | Burning fossil fuels (homes, industries, automobiles), biomass burning |
| CH₄ | Marshes, paddy fields, cattle sheds, biogas plants |
| CFCs | Refrigerators, air conditioners |
| N₂O₅ | Organic matter, fertilizers, denitrifying bacteria, automobiles |
Effects of Global Warming
- Melting of glaciers and polar ice sheets
- Rise in sea levels
- Increase in heat-related diseases
- Growth of parasites and pests
- Increased precipitation; decreased soil moisture
- Extinction of plant and animal species
The atmospheric temperature increased by 0.6°C in the 20th century due to greenhouse gas accumulation.
Preventive Measures
- Minimize fossil fuel usage
- Use energy-efficient devices
- Use recyclable and reusable products
- Reuse home wastes, newsprint, and cardboards
- Plant more trees (afforestation)
Acid Rain
Acid rain forms when sulphur oxides and nitrogen oxides emitted from power stations, factories, smelters, and automobile exhausts react with rain water in the atmosphere, forming sulphuric acid and nitric acid. If rain falls through polluted air, its acidity increases (pH drops below normal).
Effects of Acid Rain
- Damage to plants and animals
- Damage to soil chemistry
- Harm to aquatic life
- Corrosion of heritage buildings and metals
- Disruption of chemical balance in soil
Ozone Depletion
The Ozone Layer
Ozone (O₃) is a triatomic form of oxygen found in the stratosphere at about 18–50 km above Earth. It is formed and maintained in a dynamic equilibrium:
O₂ →(UV)→ [O] + [O] (dissociation)
2O₂ + 2[O] → 2O₃ (ozone formation)
The ozone layer (also called the ozone blanket) acts as a shield, absorbing harmful short-wavelength UV radiations from the sun.
Causes of Ozone Depletion
Chlorofluorocarbons (CFCs) are the primary cause. CFCs are used in refrigerators, air conditioners, fire extinguishers, and aerosol sprayers. When released, they produce active chlorine radicals (Cl and ClO) in the presence of UV radiation, which destroy ozone through chain reactions:
Ozone → Oxygen (O₂) [ozone destroyed, not replenished at the same rate]
Other contributing agents: Methane (CH₄) and oxides of nitrogen (NOₓ).
Ozone hole = A decline in the thickness of ozone over a restricted area. First discovered over Antarctica in 1985.
Effects of Ozone Depletion
More UV radiation reaching Earth causes:
- Skin cancer in humans
- Eye damage and increased incidence of cataract
- Weakening of the immune system (lowered disease resistance)
International Response
In 1987, the United Nations Environment Programme (UNEP) forged the Montreal Protocol an international agreement to freeze CFC production at 1986 levels, marking a global step toward ozone layer protection.

Class 10 Science Chapter 13 Our Environment Solved Examples
Section A: Short Answer Examples
Example 1: Name the trophic levels in the food chain: Grass → Grasshopper → Frog → Snake → Eagle.
Solution:
- Grass → 1st trophic level (Producer)
- Grasshopper → 2nd trophic level (Primary Consumer / Herbivore)
- Frog → 3rd trophic level (Secondary Consumer)
- Snake → 4th trophic level (Tertiary Consumer)
- Eagle → 5th trophic level (Quaternary Consumer / Top Carnivore)
Example 2: If 10,000 J of energy is available at the producer level, how much energy will be available at the tertiary consumer level?
Solution: Using the 10% Law:
- Producers: 10,000 J
- Primary consumers (10%): 1,000 J
- Secondary consumers (10%): 100 J
- Tertiary consumers (10%): 10 J
Example 3: Identify producers, herbivores, and carnivores: Phytoplankton → Zooplankton → Small fish → Large fish.
Solution:
- Phytoplankton = Producer
- Zooplankton = Herbivore (Primary Consumer)
- Small fish = Carnivore (Secondary Consumer)
- Large fish = Carnivore (Tertiary Consumer)
Example 4: Why can't the pyramid of energy be inverted?
Solution: Because energy always decreases progressively from one trophic level to the next, as 80–90% is lost as heat at every transfer. No trophic level can have more energy than the level below it.
Example 5: DDT concentration in water is 0.02 ppm. After passing through phytoplanktons, zooplanktons, and fish, what trend is observed?
Solution: Concentration increases at each trophic level:
- Water: 0.02 ppm → Phytoplankton/Zooplankton: 5 ppm → Fish: 240 ppm → Birds: 1600 ppm. This is biomagnification concentration multiplies with each trophic level.
Example 6: Give the photosynthesis equation showing how producers make their food.
Solution: 6CO₂ + 6H₂O →(Sunlight + Chlorophyll)→ C₆H₁₂O₆ + 6O₂
Carbon dioxide + Water → Glucose + Oxygen
Example 7: Is crow a herbivore, carnivore, or omnivore? At which trophic level does it operate?
Solution: Crow is an omnivore (double consumer). It can operate at multiple trophic levels — as a primary consumer (eating grain), secondary consumer (eating insects), or even tertiary consumer.
Example 8: A grassland has: Grass, Rabbits, Foxes, and Eagles. Draw the food chain and identify trophic levels.
Solution: Grass → Rabbit → Fox → Eagle
- Grass: 1st trophic level (Producer)
- Rabbit: 2nd trophic level (Herbivore)
- Fox: 3rd trophic level (Carnivore)
- Eagle: 4th trophic level (Top Carnivore)
Example 9: What is the main cause of acid rain and what is its effect on aquatic life?
Solution: Sulphur oxides and nitrogen oxides from power stations and vehicles react with rain water, forming sulphuric and nitric acids. Acid rain lowers the pH of water bodies, harming or killing fish and other aquatic organisms that cannot survive in highly acidic conditions.
Example 10: Why should kulhads not be used as a mass replacement for plastic cups in trains?
Solution: Although kulhads (clay cups) are biodegradable, mass production would require enormous quantities of clay, leading to the destruction of fertile topsoil which is a non-renewable resource. This environmental cost outweighs the benefit.
Section B: Application-Based Examples
Example 11: Why are top carnivores (e.g., tigers) most affected by pesticide pollution even though they don't eat crops?
Solution: Through biomagnification, pesticides like DDT accumulate and multiply in concentration at each trophic level. Tigers eat herbivores that have already concentrated pesticides from plant material. By the time pesticides reach the top carnivore level, concentrations are thousands of times higher than in the original source.
Example 12: How does using CFCs in refrigerators contribute to increased skin cancer rates?
Solution: CFCs released from refrigerators rise to the stratosphere and produce active chlorine radicals under UV radiation. These radicals destroy ozone (O₃) through chain reactions, thinning the ozone layer. With less ozone to absorb UV radiation, more UV reaches Earth's surface — directly causing skin cancer.
Example 13: In a forest ecosystem, one tree supports 500 insects, and 5 birds feed on those insects. Draw the pyramid of number. Is it upright or inverted?
Solution:
- Tree (Producer): 1
- Insects (Primary consumers): 500
- Birds (Secondary consumers): 5
This gives an intermediate (spindle-shaped) pyramid not upright, not inverted. The number first increases then decreases from base to apex.
Example 14: Calculate the energy available to a lion if 5000 J of solar energy falls on grass.
Solution:
- Grass traps 1% of solar energy: 5000 × 1/100 = 50 J
- Deer (herbivore) gets 10% of 50 J = 5 J
- Lion (carnivore) gets 10% of 5 J = 0.5 J
The lion has 0.5 J of energy available.
Example 15: Classify the following as biodegradable or non-biodegradable: (a) DDT, (b) vegetable peels, (c) uranium, (d) cloth, (e) mercury.
Solution:
- (a) DDT → Non-biodegradable
- (b) Vegetable peels → Biodegradable
- (c) Uranium → Non-biodegradable
- (d) Cloth → Biodegradable
- (e) Mercury → Non-biodegradable
Example 16: Why is the ozone hole first discovered over Antarctica and not near industrial areas?
Solution: CFCs, once released, are stable molecules that rise slowly into the stratosphere. Atmospheric circulation patterns carry them toward polar regions over time. Extremely cold temperatures over Antarctica create polar stratospheric clouds that accelerate CFC-driven ozone destruction, making the depletion most pronounced over Antarctica.
Example 17: Explain why converting biodegradable waste into biogas is considered an eco-friendly waste management method.
Solution: Biodegradable waste is fed into biogas plants where anaerobic bacteria decompose it, producing methane-rich biogas (clean fuel) and slurry/manure (natural fertilizer). This reduces landfill waste, prevents methane from escaping into the atmosphere, provides cheap energy, and returns nutrients to the soil — making it an environmentally sustainable closed-loop system.
Example 18: A student claims that flow of matter in an ecosystem is also unidirectional like energy. Is this correct? Justify.
Solution: Incorrect. Flow of matter is cyclic, not unidirectional. Elements like carbon, nitrogen, and phosphorus are continuously recycled between living organisms and the environment through biogeochemical cycles. Decomposers play a key role in returning nutrients from dead matter back to soil and air for reuse by producers. Only energy flow is unidirectional.
Example 19: Why do fewer organisms exist at higher trophic levels in a food chain?
Solution: Due to the 10% Law of Energy Transfer, only 10% of available energy passes from one trophic level to the next. As trophic levels increase, less and less energy is available to support organisms. Therefore, a large biomass of producers supports far fewer herbivores, which in turn support even fewer carnivores, creating the characteristic pyramid shape.
Example 20: Suggest three practical steps a student can take in daily life to reduce environmental damage, linking each to a specific topic from this chapter.
Solution:
- Avoid single-use plastics → Reduces non-biodegradable waste accumulation (Solid Waste Management)
- Use public transport or cycling → Reduces CO₂ and NOₓ emissions, slowing global warming and reducing acid rain (Greenhouse Effect / Acid Rain)
- Avoid aerosol spray products containing CFCs → Helps prevent ozone layer depletion (Ozone Depletion)
Our Environment Quick Revision Summary
| Topic | Key Fact |
|---|---|
| Ecosystem | Living + Non-living components interacting in biosphere |
| Producers | Green plants + blue-green algae; photoautotrophs |
| 10% Law | Proposed by Lindeman (1942); only 10% energy transferred per level |
| Decomposers | Bacteria + Fungi; essential for nutrient recycling |
| Biomagnification | DDT: 0.02 ppm (water) → 1600 ppm (birds) |
| Ozone hole | First discovered over Antarctica (1985) |
| CFC ban | UNEP Montreal Protocol (1987) |
| Greenhouse gases | CO₂, CH₄, CFCs, N₂O₅ |
| Pyramid of energy | Always upright; never inverted |
| Pyramid of biomass | Inverted in pond ecosystem |
| Minamata disease | Mercury poisoning via biomagnification |
| Acid rain | pH below normal; caused by SO₂ + NOₓ |
