Class 10 Science Chapter 3 Metals and Non-Metals Notes: Class 10 Science chapter 3 metals and non-metals notes help students understand the basic properties, uses, and reactions of different elements found in our daily life. In this chapter, we study the physical properties like hardness, malleability, ductility, conductivity, and lustre of metals, and compare them with non-metals. We also learn about chemical properties such as reactions with oxygen, water, acids, and bases, formation of ionic compounds, and the concept of valency and electron transfer.
These class 10 metals and non-metals notes are prepared according to the latest syllabus and follow the structure of metals and non metals class 10 notes ncert. Students can also look for metals and non metals class 10 notes pdf download options to revise easily before exams. A well-structured metals and non metals notes pdf provides clear explanation of important topics like corrosion, reactivity series, extraction of metals, and properties of ionic compounds.
This chapter builds a strong foundation for higher chemistry concepts. Sometimes students think all metals are hard and all non-metals are soft, but that is not always true. Understanding these small differences is very important for board exam preparation and conceptual clarity.
What is Metals and Non-Metals?
Scientists have so far discovered more than 118 elements, which are broadly classified into three categories: metals, non-metals, and metalloids.
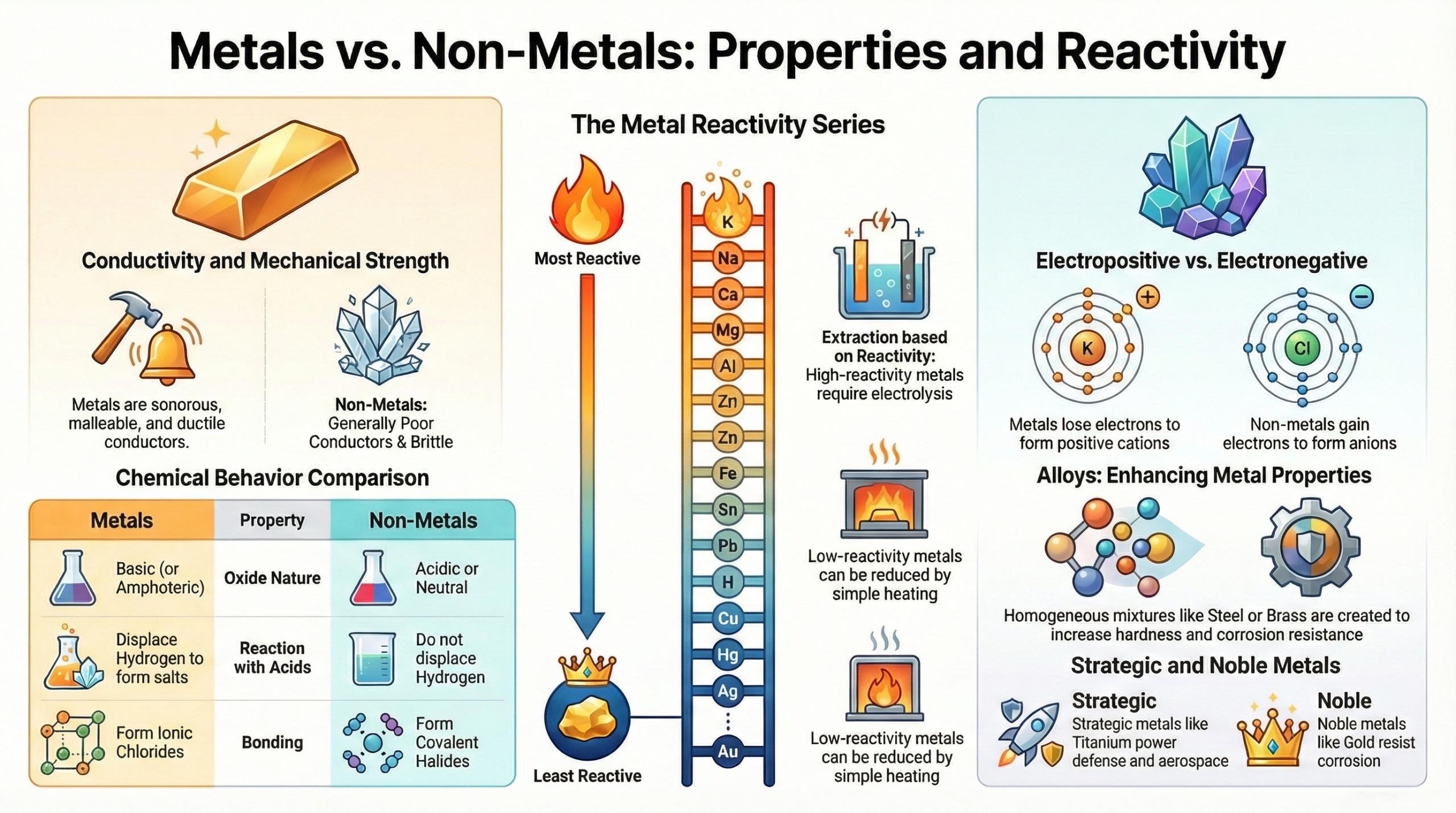
- Metals are electropositive elements hard, sonorous, malleable, ductile, and excellent conductors of heat and electricity.
- Non-metals generally have the opposite characteristics: they are electronegative, poor conductors (with exceptions), non-malleable, and often exist as gases or brittle solids.
- Metalloids (such as silicon and germanium) exhibit properties intermediate between metals and non-metals and are crucial in semiconductor technology.
Understanding the distinction between metals and non-metals is foundational to chemistry, materials science, and engineering. This chapter forms a core part of the Class 10 Chemistry curriculum and provides the conceptual basis for topics like ionic bonding, electrochemistry, and industrial processes.
CBSE Class 10 Science Metals and Non-Metals Revision Notes PDF Download
Fill the form to download this PDF
Physical Properties of Metals
| Property | Description | Notable Exceptions |
|---|---|---|
| Physical State | Solid at room temperature | Mercury (Hg) is liquid |
| Lustre | Shiny surface (metallic lustre) | — |
| Conductivity | Good conductors of heat and electricity | Lead (poor heat conductor); Mercury (poor electrical conductor) |
| Hardness | Generally hard | Sodium (Na) and Potassium (K) are soft enough to cut with a knife |
| Malleability | Can be beaten into thin sheets | — |
| Ductility | Can be drawn into wires | — |
| Sonorous | Produce a ringing sound when struck | — |
| Melting/Boiling Points | Generally high | — |
| Density | High | — |
| Valency | 1–3 electrons in outermost shell | — |
Malleability and Ductility in Practice
- Aluminium foil is used to wrap food because of aluminium's exceptional malleability.
- Gold is so ductile that one gram can be drawn into a wire nearly two kilometres long.
- Silver foil is used to decorate sweets because of silver's malleability and non-toxic nature.
- Electric wires in homes and industries are drawn from copper, aluminium, or gold due to their outstanding ductility and conductivity.
Sonority
When a tin foil is bent, it produces a characteristic creaking sound known as "tin cry" a classic demonstration of the sonorous nature of metals.
Thermal and Electrical Conductivity
When a metal rod is heated at one end, the atoms gain energy and vibrate vigorously. This energy is transferred to free electrons, which move throughout the metal lattice, passing their energy to adjacent electrons and atoms. This mobile electron model also explains why metals are good electrical conductors the free electrons carry charge continuously through the metal under an applied voltage.
Special Notes on Metals
- Three magnetic metals: Iron (Fe), Cobalt (Co), and Nickel (Ni). Steel, being primarily iron, is also magnetic.
- Lightest metal: Lithium (Li)
- Heaviest metal: Iridium (Ir)
- Strategic metals like titanium, chromium, manganese, and zirconium play critical roles in aerospace, defence, and nuclear technology.
Physical Properties of Non-Metals
| Property | Description | Notable Exceptions |
|---|---|---|
| Physical State | Gas or solid at room temperature | Bromine (Br₂) is liquid |
| Surface | Dull, non-lustrous | Diamond and iodine crystals are lustrous |
| Conductivity | Poor conductors | Graphite conducts electricity |
| Hardness | Generally soft | Diamond is the hardest known natural substance |
| Malleability | Non-malleable and non-ductile | — |
| Sonorous | Not sonorous | — |
| Melting/Boiling Points | Generally low | Diamond has an exceptionally high melting point |
| Reactivity | Form acidic or neutral oxides | — |
There are only 22 non-metallic elements, of which 11 are gases, 1 is a liquid (bromine), and 10 are solids. Examples include oxygen (O₂), nitrogen (N₂), chlorine (Cl₂), carbon (C), sulphur (S₈), and phosphorus (P₄).
Chemical Properties of Metals
Metals are electropositive they lose electrons to form positive ions (cations). This fundamental tendency drives their characteristic chemical behaviour.
(I) Reaction with Oxygen
Almost all metals combine with oxygen to form metal oxides, which are basic in nature:
Metal + Oxygen → Metal Oxide
Reactivity trend with oxygen:
Na > Mg > Zn > Fe > Cu
- Sodium and Potassium catch fire spontaneously in moist air. They burn with characteristic colours (Na: golden yellow; K: pink-violet) and must be stored in kerosene.4Na + O₂ → 2Na₂O
- Magnesium reacts only on heating and can also combine with nitrogen: 2Mg + O₂ → 2MgO6Mg + 2N₂ → 2Mg₃N₂ (Magnesium nitride)
- Zinc and Iron form protective oxide layers that prevent further oxidation. 3Fe(s) + O₂(g) → Fe₃O₄(s)
- Lead, Silver, and Gold do not react with oxygen even at high temperatures these are called noble metals.
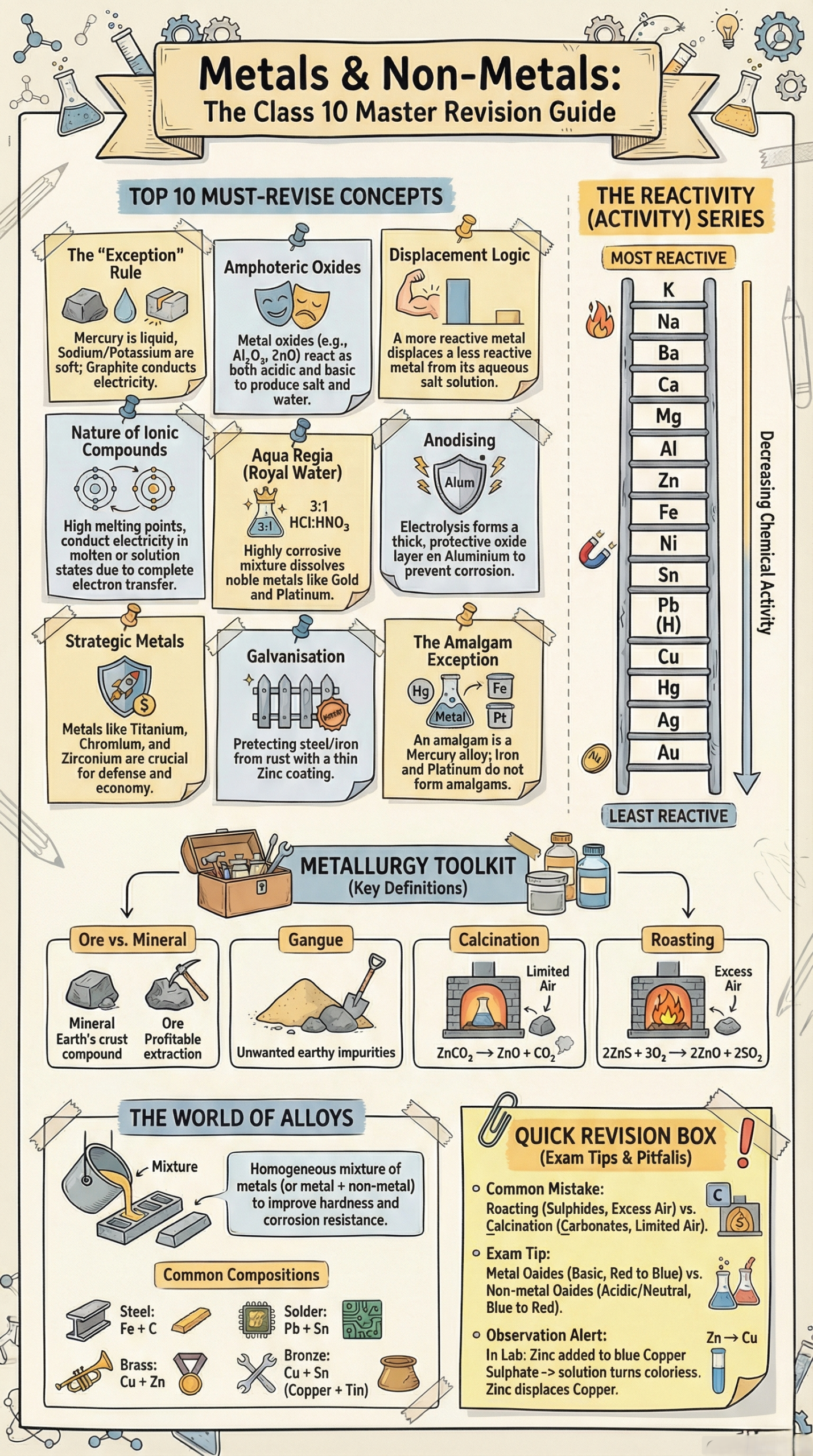
Amphoteric Oxides
Aluminium oxide (Al₂O₃) and zinc oxide (ZnO) are amphoteric they react with both acids and bases:
Al₂O₃ + 6HCl → 2AlCl₃ + 3H₂O (reacts with acid)
Al₂O₃ + 2NaOH → 2NaAlO₂ + H₂O (reacts with base)
Anodising
Anodising is an electrolytic process in which aluminium articles are made the anode in dilute H₂SO₄. The oxygen released reacts with the aluminium surface to form a thick, durable oxide layer, which can then be dyed for decorative finishes and resists further corrosion.
(II) Reaction with Water
Metal + Water → Metal Oxide + Hydrogen Metal
Oxide + Water → Metal Hydroxide
Order of reactivity with water:
K > Na > Ca > Mg > Zn > Fe > Cu
- Potassium and Sodium react violently with cold water, releasing hydrogen that immediately catches fire: 2K(s) + 2H₂O(l) → 2KOH(aq) + H₂(g) + Heat
2Na(s) + 2H₂O(l) → 2NaOH(aq) + H₂(g) + Heat
- Calcium reacts less violently; hydrogen bubbles stick to its surface causing it to float: Ca(s) + 2H₂O(l) → Ca(OH)₂(aq) + H₂(g)
- Aluminium, Iron, and Zinc react only with steam:2Al(s) + 3H₂O(g) → Al₂O₃(s) + 3H₂(g)
3Fe(s) + 4H₂O(g) ⇌ Fe₃O₄(s) + 4H₂(g)
- Lead, Copper, Silver, and Gold do not react with water at all.
(III) Reaction with Acids
When metals react with dilute acids, a salt and hydrogen gas are produced:
Metal + Dilute Acid → Metal Salt + Hydrogen
Reactivity order:
Na > Mg > Zn > Fe > Cu
Key examples:
2Na(s) + 2HCl(aq) → 2NaCl(aq) + H₂(g) [explosive]
Mg(s) + 2HCl(aq) → MgCl₂(aq) + H₂(g)
Zn(s) + 2HCl(aq) → ZnCl₂(aq) + H₂(g)
Fe(s) + 2HCl(aq) → FeCl₂(aq) + H₂(g)
Cu(s) + HCl(aq) → No reaction
Important: Hydrogen is not evolved when metals react with nitric acid (HNO₃) because HNO₃ is a strong oxidising agent it oxidises the H₂ to water. Exceptions: Mg and Mn react with very dilute HNO₃ to produce H₂.
Aqua Regia
Aqua regia (Latin for "royal water") is a mixture of 3 parts concentrated HCl and 1 part concentrated HNO₃. It is the only acid that can dissolve noble metals like gold and platinum:
Au + 3Cl → AuCl₃ (Auric chloride) Pt + 4Cl → PtCl₄ (Platinic chloride)
(IV) Reaction with Chlorine
Most metals react with chlorine to form ionic chlorides:
2Na(s) + Cl₂(g) → 2NaCl(s)
Mg(s) + Cl₂(g) → MgCl₂(s)
Zn(s) + Cl₂(g) → ZnCl₂(s)
During these reactions, the metal is oxidised (loses electrons) and chlorine is reduced (gains electrons).
(V) Reaction with Hydrogen
Most metals do not combine with hydrogen. However, a few highly reactive metals form ionic hydrides:
2Na(s) + H₂(g) → 2NaH(s) (Sodium hydride)
2K(s) + H₂(g) → 2KH(s) (Potassium hydride)
Ca(s) + H₂(g) → CaH₂(s) (Calcium hydride)
(VI) Displacement Reactions – Metals and Salt Solutions
A more reactive metal displaces a less reactive metal from its salt solution:
Metal A + Salt solution of B → Salt solution of A + Metal B
Example:
Zn(s) + CuSO₄(aq) → ZnSO₄(aq) + Cu(s)
Fe(s) + CuSO₄(aq) → FeSO₄(aq) + Cu(s)
This principle underpins electrochemical cells and is used to test relative reactivity.
Activity Series of Metals
The Activity Series (also called the Reactivity Series) arranges metals in decreasing order of reactivity:
K > Na > Ba > Ca > Mg > Al > Zn > Fe > Ni > Sn > Pb > H > Cu > Hg > Ag > Au
Significance of the Activity Series
- Metals above hydrogen can displace H₂ from dilute acids.
- A more electropositive metal replaces a less electropositive metal from its salt solution.
- Metals at the top (K, Na, Ca, Mg, Al) are extracted by electrolysis.
- Metals in the middle (Zn, Fe, Pb) are extracted by carbon reduction.
- Metals at the bottom (Cu, Hg, Ag) are extracted by heat alone.
- Metals at the very bottom (Pt, Au) are found in the native (free) state.
Reaction of Metals with Non-Metals – Ionic Bonding
When metals react with non-metals, electrons transfer from the metal to the non-metal, forming ionic compounds. The metal becomes a cation and the non-metal becomes an anion.
Example – Sodium Chloride (NaCl):
Na (2,8,1) → Na⁺ (2,8) + e⁻ Cl (2,8,7) + e⁻ → Cl⁻ (2,8,8) Na⁺ + Cl⁻ → NaCl (ionic compound)
Example – Magnesium Chloride (MgCl₂):
Mg (2,8,2) → Mg²⁺ (2,8) + 2e⁻ 2Cl + 2e⁻ → 2Cl⁻ Mg²⁺ + 2Cl⁻ → MgCl₂
Properties of Ionic Compounds
| Property | Detail |
|---|---|
| Physical Nature | Solid and somewhat hard |
| Melting/Boiling Points | High (strong electrostatic forces) |
| Solubility | Generally soluble in water; insoluble in kerosene, petrol |
| Electrical Conductivity | Non-conductors in solid state; conductors in molten state or aqueous solution |
Occurrence of Metals in Nature
Metals occur in nature in two forms:
- Free (Native) State: Metals unreactive to moisture, O₂, and CO₂ — e.g., Gold, Platinum, Silver, Mercury.
- Combined State: Reactive metals found as compounds (oxides, carbonates, sulphides, chlorides) e.g., Sodium, Calcium, Aluminium, Copper.
Minerals, Ores, and Gangue
- Mineral: Any naturally occurring solid compound or element found in the Earth's crust.
- Ore: A mineral from which a metal can be profitably and economically extracted. All ores are minerals, but not all minerals are ores.
- Gangue (Matrix): Unwanted impurities (sand, rock, clay) mixed with the ore.
Common Ores
| Metal | Ore Name | Formula |
|---|---|---|
| Iron | Haematite | Fe₂O₃ |
| Iron | Magnetite | FeO·Fe₂O₃ |
| Aluminium | Bauxite | Al₂O₃·2H₂O |
| Copper | Copper Pyrites | CuFeS₂ |
| Zinc | Zinc Blende | ZnS |
| Lead | Lead Glance (Galena) | PbS |
| Mercury | Cinnabar | HgS |
| Calcium | Limestone | CaCO₃ |
| Sodium | Rock Salt | NaCl |
| Silver | Horn Silver | AgCl |
Metallurgy – Extraction of Metals
Metallurgy is the science and technology of extracting metals from their ores and refining them for practical use.
Steps in Metallurgy
- Enrichment (Concentration/Dressing) of Ore
- Conversion of enriched ore into metal oxide (Calcination or Roasting)
- Extraction of metal from metal oxide (Reduction)
- Refining (Purification) of metal
Concentration of Ores
The process of removing gangue from ore is called dressing, concentration, or beneficiation.
Methods of Concentration
I. Hand Picking Used when ore and gangue particles differ significantly in size and appearance. Example: Haematite ore.
II. Hydraulic Washing (Gravity Separation / Levigation) Based on differences in specific gravity. A stream of water washes away lighter gangue particles, leaving behind denser ore particles. Used for oxide ores of iron (haematite), tin (tinstone), gold, and silver.
III. Magnetic Separation Applied when either the ore or the gangue is magnetic. The powdered ore is dropped onto a moving belt over magnetic rollers magnetic particles are attracted and separated from non-magnetic particles. Example: Magnetite (magnetic ore of iron); tungstate impurities removed from tin stone.
IV. Froth Flotation Used primarily for sulphide ores (copper pyrites, galena, zinc blende). The ore is mixed with water and pine oil. Compressed air produces froth. Sulphide ore particles are wetted by oil and rise with the froth; gangue is wetted by water and sinks. The ore-laden froth is skimmed off and washed.
V. Leaching (Chemical Separation) Based on differences in chemical reactivity. The ore is treated with a suitable reagent that selectively dissolves the ore. Example: Baeyer's process - Bauxite is treated with concentrated NaOH:
Al₂O₃(s) + 2NaOH(aq) + H₂O → 2NaAlO₂ + 2H₂O (Sodium aluminate) 2NaAlO₂(aq) + 2H₂O → Al(OH)₃(s)↓ + 2NaOH 2Al(OH)₃ → Al₂O₃ + 3H₂O (at 1473K)
Calcination vs. Roasting
| Feature | Calcination | Roasting |
|---|---|---|
| Air supply | Limited | Excess |
| Used for | Carbonate/hydrate ores | Sulphide ores |
| Result | Carbonate → Oxide + CO₂ | Sulphide → Oxide + SO₂ |
| Example | ZnCO₃ → ZnO + CO₂ | 2ZnS + 3O₂ → 2ZnO + 2SO₂ |
Both processes are carried out in a reverberatory furnace.
Chemical Reduction Methods
(A) Reduction by Carbon (Smelting)
The roasted/calcined ore is mixed with coke (carbon) and heated above its melting point. Carbon reduces the metal oxide:
ZnO(s) + C(s) → Zn(s) + CO(g)
SnO₂(s) + 2C(s) → Sn + 2CO(g)
Carbon monoxide produced can also reduce oxides:
Fe₂O₃ + CO → 2FeO + CO₂
FeO + CO → Fe + CO₂
The most important industrial application is iron extraction in a blast furnace.
(B) Reduction by Hydrogen
Used for metals like Tungsten (W) and Molybdenum (Mo) that cannot be reduced by carbon:
WO₃ + 3H₂ → W + 3H₂O
Hydrogen is rarely used due to high cost and flammability.
(C) Aluminothermy (Thermite Reaction)
Aluminium powder is used to reduce metal oxides that cannot be easily reduced by carbon (e.g., Cr₂O₃, MnO₂):
Cr₂O₃(s) + 2Al(s) → Al₂O₃(s) + 2Cr(l)
Fe₂O₃(s) + 2Al(s) → Al₂O₃(s) + 2Fe(l) [Thermite reaction]
The thermite reaction is highly exothermic the molten iron produced is used in welding railway tracks and repairing cracked machine parts.
(D) Reduction by Magnesium
Used for metals like titanium:
TiCl₄ + 2Mg → 2MgCl₂ + Ti
(E) Electrolytic Reduction
Highly reactive metals (K, Na, Ca, Mg, Al) that cannot be reduced by chemical means are extracted by electrolysis of their molten salts (halides):
- Sodium from molten NaCl:At cathode: 2Na⁺ + 2e⁻ → 2NaAt anode: 2Cl⁻ → Cl₂ + 2e⁻
- Aluminium from Al₂O₃ dissolved in molten cryolite (Na₃AlF₆) using graphite electrodes:At cathode: Al³⁺ + 3e⁻ → Al(l)At anode: 2O²⁻ → O₂(g) + 4e⁻
Refining of Metals
The purification of crude metal is called refining. Methods used depend on the nature of the metal and its impurities.
| Method | Principle | Metals Refined |
|---|---|---|
| Distillation | Boiling point difference | Zn, Hg, Cd |
| Liquation | Melting point difference | Bi, Sn, Pb |
| Bassemerisation | Oxidation of impurities | Cu, Ag, Sn |
| Cupellation | Oxidation; removes Pb from Ag | Ag |
| Poling | Hydrocarbon gases reduce metal oxide impurities | Cu (blister copper) |
| Electro-refining | Electrolysis; impure metal = anode, pure metal strip = cathode | Cu, Au, Ag, Pb, Zn, Al |
| Zone Refining | Impurities concentrate in molten zone | Ga, In, Si (semiconductors) |
| Mond Process | Volatile nickel carbonyl formation and decomposition | Ni |
| Van-Arkel Method | Volatile iodide formation and decomposition | Zr, Ti |
Electro-Refining (Detail)
- Impure metal acts as the anode
- Pure metal strip acts as the cathode
- Electrolyte is a soluble salt of the same metal
- On passing current: metal from anode dissolves → deposits as pure metal on cathode
- Impurities either remain in solution or settle as anode mud (e.g., Au, Ag)
Alloys – Types, Properties and Uses
An alloy is a homogeneous mixture of two or more metals, or a metal and a non-metal. An alloy containing mercury is called an amalgam.
Methods of Preparation
- Melting components together in the correct proportions and cooling
- Compressing components under high pressure
- Simultaneous electrodeposition of metals
Properties of Alloys
- Harder than individual constituent metals, but less ductile and malleable
- More resistant to corrosion
- Melting point may differ from constituents
- Electrical conductivity is generally lower than pure metals
- Properties are greatly improved compared to individual metals
Important Alloys and Their Uses
Alloys of Aluminium
| Alloy | Composition | Uses |
|---|---|---|
| Magnallium | Al 95%, Mg 5% | Pressure cookers, balance beams |
| Duralumin | Al 95%, Cu 4%, Mg 0.5%, Mn 0.5% | Aircraft parts, automobiles |
Alloys of Copper
| Alloy | Composition | Uses |
|---|---|---|
| Bronze | Cu 90%, Sn 10% | Statues, coins, utensils |
| Brass | Cu 80%, Zn 20% | Utensils, machinery, wires |
| Gun Metal | Cu 90%, Sn 10% | Gun barrels |
| Bell Metal | Cu 80%, Sn 20% | Bells and gongs |
| German Silver | Cu 60%, Zn 20%, Ni 20% | Silver wares, resistance wires |
| Phosphor Bronze | Cu 95%, Sn 4.8%, P 0.2% | Springs, electric switches |
Alloys of Iron (Alloy Steels)
| Alloy | Composition | Uses |
|---|---|---|
| Stainless Steel | Fe 73%, Cr 18%, Ni 8%, C 1% | Utensils, surgical instruments, watch cases |
| Nickel Steel | Fe 96–98%, Ni 2–4% | Cables, armour plates, gears |
| Alnico | Fe 60%, Ni 20%, Al 12%, Co 8% | Permanent magnets |
| Chrome Steel | Fe 98%, Cr 2% | Axles, ball bearings, cutting tools |
Alloys of Silver
| Alloy | Composition | Uses |
|---|---|---|
| Coinage Silver | Ag 90%, Cu 10% | Coins |
| Dental Alloy | Ag 33%, Hg 52%, Sn 12.5%, Cu 2% | Dental fillings |
Gold Carats
- 24 carat gold = pure gold (100% gold; too soft for ornaments)
- 22 carat gold = 22 parts gold + 2 parts silver or copper in 24 parts of alloy
- 18 carat gold = 18 parts gold + 6 parts copper/silver in 24 parts
Corrosion and Rusting
What is Corrosion?
Corrosion is the slow, gradual destruction of a metal due to chemical reactions on its surface with oxygen, moisture, carbon dioxide, sulphur dioxide, or hydrogen sulphide from the atmosphere.
In corrosion, metal atoms are oxidised they lose electrons and form ions:
M → M⁺ + e⁻
Familiar Examples
- Silver turns black due to formation of silver sulphide (Ag₂S) on reaction with atmospheric sulphur.
- Copper develops a green coating of basic copper carbonate [Cu(OH)₂·CuCO₃] when exposed to moist CO₂.
- Iron develops reddish-brown rust [Fe₂O₃·xH₂O + Fe(OH)₃].
Rusting of Iron
Rusting is an oxidation reaction requiring both water and oxygen simultaneously. Air-free water and dry oxygen alone do not cause rusting.
Fe + H₂O + O₂ → Fe₂O₃·xH₂O + Fe(OH)₃
Factors that Accelerate Corrosion
- Contact between two dissimilar metals: Forms a galvanic cell; the more reactive metal corrodes faster (e.g., iron corrodes faster when in contact with copper).
- Atmospheric pollutants: CO₂, SO₂, H₂S from industrial emissions accelerate corrosion significantly.
Prevention of Rusting
| Method | Description |
|---|---|
| Barrier Protection | Coating with paint, grease, enamel, or oil |
| Galvanisation | Coating iron/steel with a thin layer of zinc; zinc self-forms a protective basic zinc carbonate [Zn(OH)₂·ZnCO₃] |
| Electroplating | Coating with chromium, nickel, tin, or aluminium |
| Alloying | Making stainless steel (iron + chromium + nickel) |
| Oxide Coating | Coating surface with Fe₃O₄ (iron II, III oxide) |
| Sacrificial Protection | Connecting more reactive metals (Mg or Zn) to iron; the more reactive metal oxidises preferentially, protecting iron |
Chemical Properties of Non-Metals
Non-metals are electronegative — they gain electrons to form anions:
Cl + e⁻ → Cl⁻ O + 2e⁻ → O²⁻ S + 2e⁻ → S²⁻
(I) Reaction with Oxygen
Non-metals form acidic or neutral oxides (never basic):
C(s) + O₂(g) → CO₂(g) S(s) + O₂(g) → SO₂(g)
These dissolve in water to form acids:
CO₂ + H₂O → H₂CO₃ (Carbonic acid) SO₂ + H₂O → H₂SO₃ (Sulphurous acid)
P₂O₅ + 3H₂O → 2H₃PO₄ (Phosphoric acid)
Neutral oxides (no effect on litmus): CO, N₂O
(II) Non-Metals Do NOT React with Dilute Acids
Non-metals cannot supply electrons to H⁺ ions, so they cannot displace hydrogen from dilute acids.
(III) Reaction with Chlorine
Non-metals form covalent chlorides (volatile liquids or gases):
H₂ + Cl₂ → 2HCl P₄ + 6Cl₂ → 4PCl₃ P₄ + 10Cl₂ → 4PCl₅
(IV) Reaction with Hydrogen
Non-metals combine with hydrogen to form stable covalent hydrides:
N₂ + 3H₂ → 2NH₃ (Ammonia)
H₂ + S → H₂S (Hydrogen sulphide)
2H₂ + O₂ → 2H₂O (Water)
Important Compounds of Non-Metals
Ozone (O₃)
An allotrope of oxygen and a powerful oxidising agent. The ozone layer in the stratosphere absorbs harmful UV radiation from the sun, protecting life on Earth.
Industrial uses: bleaching agent, disinfectant, purification of drinking water, production of artificial silk.
Hydrogen Peroxide (H₂O₂)
Structure: H–O–O–H. Exhibits both oxidising and reducing properties.
Uses:
- Disinfectant for wounds (dilute solution)
- Bleaching silk, wool, ivory, and hair
- Rocket fuel oxidant (concentrated)
- Restoring old lead-based paintings by converting black PbS back to white PbSO₄
Ammonia (NH₃)
Pyramidal structure. Key uses:
- Manufacturing nitrogen fertilisers (urea, ammonium nitrate, ammonium sulphate)
- Manufacturing nitric acid (Ostwald's process)
- Refrigerant in ice factories
- Removing fat and grease stains
Nitric Acid (HNO₃)
A strong oxidising agent. Uses:
- Production of fertilisers (ammonium nitrate)
- Manufacturing explosives (TNT, nitroglycerine)
- Purification of gold and silver
- Manufacturing dyes, medicines, and sulphuric acid
Sulphuric Acid (H₂SO₄) – Also known King of Chemicals
The most widely manufactured industrial chemical. Uses:
- Manufacturing HCl, HNO₃, and fertilisers
- Lead-acid batteries
- Petroleum refining
- Dye, detergent, and electroplating industries
- Metallurgy
Differences between Metals vs. Non-Metals
| Property | Metals | Non-Metals |
|---|---|---|
| Reaction with acids | Displace H₂ | Do not displace H₂ |
| Nature of oxides | Basic (some amphoteric) | Acidic or neutral |
| Hydrides | Few, ionic | Stable, covalent |
| Halides | Ionic, crystalline solids | Covalent, volatile |
| Electrochemical behaviour | Electropositive; form cations; deposit at cathode | Electronegative; form anions; liberate at anode |
| Redox role | Reducing agents | Oxidising agents |
Points to Remember
- Metalloids have intermediate properties between metals and non-metals.
- Allotropes are different structural forms of the same element with the same chemical properties but different physical properties.
- Metal oxides are basic; non-metal oxides are acidic or neutral; some metal oxides (Al₂O₃, ZnO) are amphoteric.
- Aqua regia = 3 parts conc. HCl + 1 part conc. HNO₃; dissolves gold and platinum.
- The activity series helps predict which metals can displace others and which extraction method to use.
- Ionic compounds conduct electricity only in molten state or when dissolved in water.
- Galvanisation coats iron with zinc to prevent rusting; zinc forms its own protective oxide layer.
- An alloy is a homogeneous mixture; an amalgam contains mercury as one component.
- Aluminium is the most abundant metal in Earth's crust; its ore is bauxite.
- Roasting converts sulphide ores to oxides; calcination converts carbonate ores to oxides.
