Electricity is one of the most fundamental topics in Class 10 Physics formulas , forming the backbone of modern technology and engineering. Whether you are a student preparing for CBSE board exams or a curious learner exploring the physical world, understanding electricity - from the static charge on a comb to the current flowing through your home circuits is essential. This comprehensive guide covers every concept with clarity, precision, and exam-focused depth.
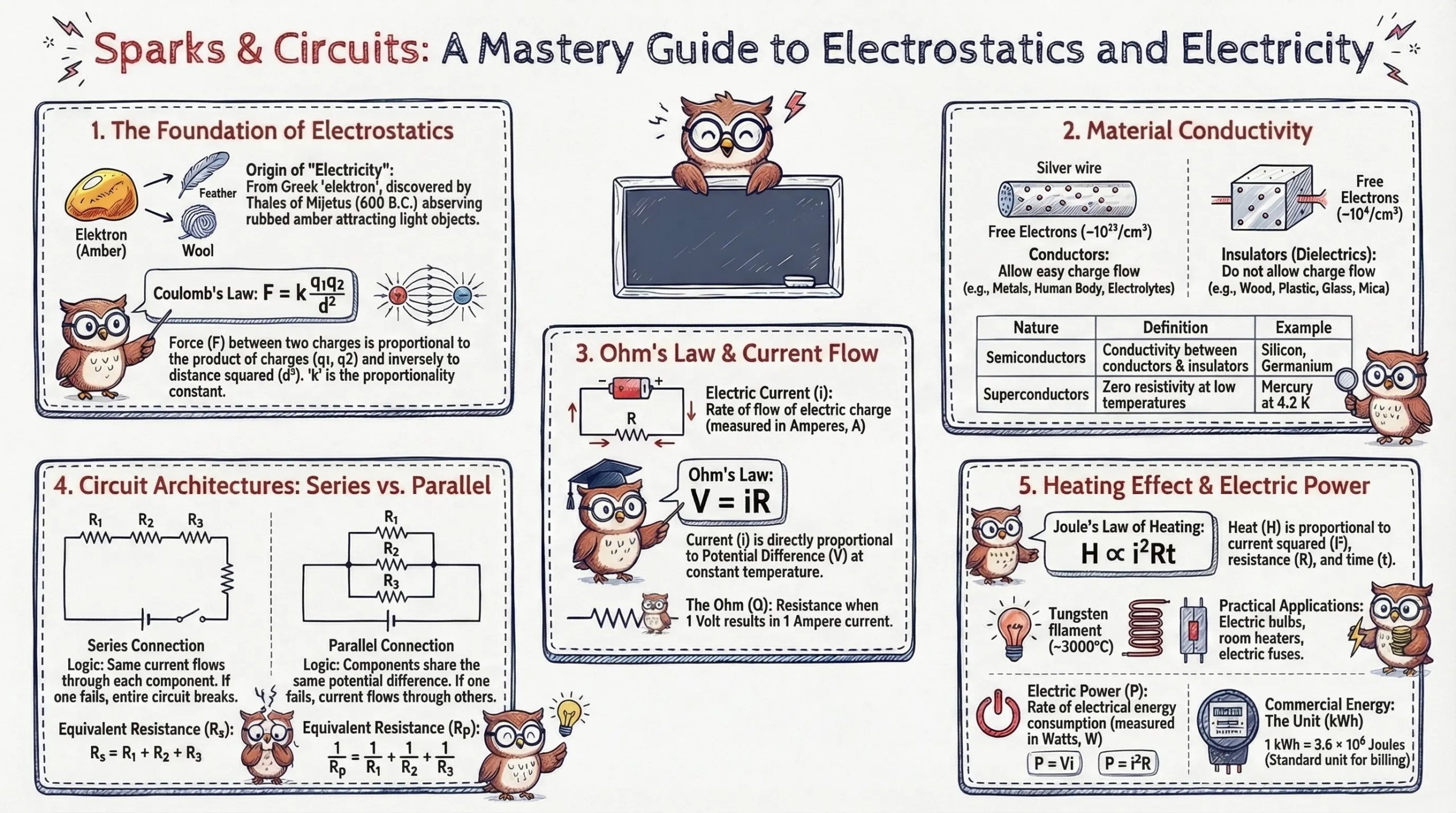
Historical Background of Electricity
The story of electricity begins over 2,600 years ago, with observations that still underpin our modern understanding.
| Scientist | Period | Contribution |
|---|---|---|
| Thales of Miletus | ~600 B.C. | Discovered that rubbed amber attracts light objects the first recorded electrical phenomenon. The Greek word for amber, elektron, gave us the word "electricity." |
| Dr. William Gilbert | 1540–1603 A.D. | Wrote De Magnate, the first systematic scientific study of electrical and magnetic phenomena. Introduced the terms "electric force," "electric attraction," and "magnetic pole." |
| C.F. Du Fay | 1698–1739 A.D. | Proposed that two kinds of electricity exist "vitreous" (positive) and "resinous" (negative) and formulated the Two Fluid Theory of Electricity. |
| Benjamin Franklin | 1747 | Proposed the One-Fluid Model of Electricity, introducing the terms "positive" and "negative" charge. By this convention, the electron carries a negative charge. |
Expert Insight: Thales' observation was not mere curiosity it laid the conceptual foundation for electrostatics, the branch of physics dealing with stationary electric charges.
CBSE Class 10 Physics Notes - Electricity Revision Notes PDF
Fill the form to download this PDF
Electrification - Types and Mechanisms
Electrification is the process of charging a body. Since electrons are highly mobile and relatively easy to remove, all electrification fundamentally involves electron transfer.
- Excess of electrons → Negative charge
- Deficiency of electrons → Positive charge
When charge does not flow but remains static on a body, it is called Static Electricity. The branch of physics studying stationary charges is Electrostatics.
Types of Electrification
(a) Frictional Electrification
When two insulating bodies are rubbed against each other, electrons transfer from one to the other, generating equal and opposite charges on each body.
Real-World Examples:
- Spark at a metal knob: Walking across a carpet causes frictional electrification. Electrons migrate from the carpet to your body. Touching a metal doorknob causes these electrons to discharge rapidly, producing a shock.
- Hair-raising experience: Removing a woollen hat from your head transfers electrons from hair to hat, leaving hair positively charged. Since like charges repel, each hair strand pushes away from the others literally making your hair stand up.
Note: Static electricity is more noticeable in winter because dry air is a poor conductor. In summer, atmospheric humidity helps electrons dissipate quickly, preventing significant charge build-up.
(b) Charging by Conduction
Charging by conduction occurs when a charged body directly contacts an uncharged conductor, causing electrons to flow between them.
- If the charging body is positive: it attracts electrons from the uncharged body → uncharged body becomes positively charged.
- If the charging body is negative: it transfers electrons to the uncharged body → uncharged body becomes negatively charged.
Positive charges cannot move between bodies. Only electrons (negative charges) are transferred. A body appears "positively charged" simply because it has lost electrons.
(c) Electrostatic Induction
Induction is the process of charging a conductor by bringing a charged body near it without physical contact. The charges that appear on the conductor are called induced charges.
Mechanism of Induced Charge Separation:
- When a negatively charged rod is brought near a metallic ball, free electrons in the ball are repelled to the far side. The near side becomes positively charged.
- When a positively charged rod approaches, free electrons are attracted toward the near side, leaving the far side positively charged.
- In both cases, the charge induced on the near side is opposite to the charge on the approaching rod.
Charging Two Spheres by Induction:
- Place two neutral spheres (A and B) in contact on insulating stands.
- Bring a negatively charged rod near sphere A.
- Electrons in both spheres migrate toward sphere B (far side), making B negative and A positive.
- Separate the spheres while the rod is still nearby.
- Remove the rod — each sphere retains its induced charge.
Charging a Single Sphere by Induction (to obtain positive charge):
- Bring a negatively charged rod near the sphere.
- Electrons migrate to the far end (right side).
- Earth (ground) the far end — electrons flow to earth.
- Remove the earth connection.
- Remove the rod — positive charge distributes uniformly over the sphere.
Key Principle: Induction precedes attraction. When a charged body attracts an uncharged conductor, the near side of the conductor develops an opposite charge (by induction), which is closer and therefore attracts more strongly than the far side repels.
Earthing a Charged Metal Sphere:
- The earth's electric potential is always taken as zero.
- If a positively charged sphere (higher potential) is earthed, electrons flow from earth to the sphere, neutralising the positive charge.
- If a negatively charged sphere (lower potential, in magnitude) is earthed, electrons flow from the sphere to earth, again neutralising the charge.
Triboelectric Series
The Triboelectric Series arranges materials in increasing order of electron affinity. When two materials are rubbed together:
- The material higher in the series (lower electron affinity) loses electrons → becomes positively charged.
- The material lower in the series (higher electron affinity) gains electrons → becomes negatively charged.
Triboelectric Series (Positive to Negative)
| Position | Material |
|---|---|
| 1 (Most positive tendency) | Fur |
| 2 | Cat's skin |
| 3 | Wax |
| 4 | Glass |
| 5 | Human hair |
| 6 | Wool |
| 7 | Cotton |
| 8 | Paper |
| 9 | Silk |
| 10 | Human body |
| 11 | Wood |
| 12 | Metals |
| 13 | India-rubber |
| 14 | Resin |
| 15 | Amber |
| 16 | Sulphur |
| 17 (Most negative tendency) | Ebonite |
Example: Glass (position 4) rubbed with Silk (position 9) → Glass loses electrons and becomes positively charged; Silk gains electrons and becomes negatively charged.
Properties of Electric Charge
Understanding the fundamental properties of electric charge is essential for mastering all of electrostatics.
Scalar Nature
Electric charge is a scalar quantity. It has magnitude but no direction. It exists in only two types: positive and negative.
Like and Unlike Charges
- Like charges repel each other.
- Unlike charges attract each other.
Additivity of Charge
The total charge on a body is the algebraic sum of all individual charges, each taken with its proper sign.
Conservation of Electric Charge
Electric charge can neither be created nor destroyed. It can only be transferred from one body to another.
- When a glass rod is rubbed with silk, the glass gains +Q charge and silk gains −Q charge. Total charge = 0 (same as before).
- Conservation of charge holds in all physical processes chemical reactions, nuclear reactions, and everyday electrical phenomena.
Quantisation of Charge
Every charge in nature is an integral multiple of the elementary charge e (charge of one electron):
q = ± ne
Where:
- n = 1, 2, 3, 4, ...
- e = 1.6 × 10⁻¹⁹ C (elementary charge)
Since electrons are indivisible, only whole numbers of electrons can be transferred. Therefore, no fractional charge exists independently in nature.
Charge is Associated with Mass
Charge cannot exist without mass. A body gaining electrons gains mass (9.11 × 10⁻³¹ kg per electron); a body losing electrons loses mass.
- Photons have no mass → they cannot carry charge.
- Alpha rays, beta rays, anode rays, and cathode rays all have mass → they carry charge.
Charge is Transferable
When a charged body contacts an uncharged conductor, electrons transfer between them (conduction).
Charge is Invariant
Electric charge does not change with the speed of the body. (Note: mass increases with speed, but charge remains constant.)
Accelerated Charge Radiates Energy
- A charge at rest produces only an electric field (E⃗,).
- A charge in uniform motion produces both electric (E⃗) and magnetic fields (B⃗).
- A charge under acceleration produces E⃗, B⃗, and radiates energy as electromagnetic waves.
Charge Resides on the Outer Surface
Like charges repel each other and move as far apart as possible. Hence, all charge on a conductor resides on its outer surface.
A solid sphere and a hollow sphere of the same outer radius hold the same maximum charge, because charge only occupies the outer surface.
Materials: Conductors, Insulators, Semiconductors & Superconductors
| Type | Definition | Reason | Examples |
|---|---|---|---|
| Conductors | Allow free flow of electric charge | Large number of free electrons (e.g., 10²³ per cm³ in silver) | Metals, human body, earth, electrolytes (acid/base/salt solutions) |
| Insulators (Dielectrics) | Do not allow charge to flow | Very few free electrons (e.g., 10⁴ per cm³ in mica) | Wood, silk, rubber, plastic, mica, wax, glass, diamond, sulphur, ebonite |
| Semiconductors | Conductivity between conductors and insulators | Charge carriers are both electrons and holes | Silicon, Germanium |
| Superconductors | Zero electrical resistance at a specific temperature | Electron pairs (Cooper pairs) are undeflected by electric and magnetic fields | Mercury at 4.2 K |
Coulomb's Law
About Charles Augustin de Coulomb (1736–1806)
Coulomb was a distinguished 18th-century French scientist who systematically studied the forces between electric charges. His findings, summarised in Coulomb's Law, form the foundation of electrostatics.
Statement of Coulomb's Law
The electrostatic force between two point charges is:
- Directly proportional to the product of the magnitudes of the two charges.
- Inversely proportional to the square of the distance between them.
- Acts along the line joining the two charges.
Mathematical Expression
For charges q₁ and q₂ separated by distance d:
F = k × (q₁ × q₂) / d²
In SI units:
k = 1 / (4πε₀) = 9 × 10⁹ N·m²/C²
Where ε₀ = 8.85 × 10⁻¹² C²/N·m² is the permittivity of free space.
In air/vacuum:
F = 9 × 10⁹ × (q₁ × q₂) / d²
In a medium with relative permittivity εᵣ:
F = (9 × 10⁹ / εᵣ) × (q₁ × q₂) / d²
Significance of Relative Permittivity (εᵣ)
Also called the dielectric constant, εᵣ tells us how much a medium reduces the electrostatic force between charges.
| Medium | Dielectric Constant (εᵣ) |
|---|---|
| Vacuum/Air | 1 |
| Paper | 3.7 |
| Mica | 3–6 |
| Pyrex Glass | 5–6 |
| Porcelain | 7 |
| Glycerin | 42.5 |
| Water (at 20°C) | 80 |
| Benzene | 2.3 |
If εᵣ for glass = K, then the force between charges in glass is K times less than the same charges in air at the same separation.
Units of Charge
- SI Unit: Coulomb (C)
- CGS Unit: Statcoulomb (1 C = 3 × 10⁹ statcoulomb)
- Smallest charge: Charge on electron = 1.602 × 10⁻¹⁹ C
- Largest practical unit: Faraday (1 F = 96,500 C)
- 1 coulomb = 6.25 × 10¹⁸ electrons
- 1 faraday = 6.023 × 10²³ electrons (Avogadro's number)
Comparison: Electrostatic vs. Gravitational Force
For two unit masses (1 kg each) and two unit charges (1 C each) separated by 1 m:
- Gravitational force: Fɡ = 6.627 × 10⁻¹¹ N
- Electrostatic force: Fₑ = 9 × 10⁹ N
Fₑ / Fɡ = 1.35 × 10²⁰
The electrostatic force is approximately 10²⁰ times stronger than the gravitational force illustrating why electrostatics dominates at the atomic and molecular scale.
Potential Difference and the Flow of Charge
Electric Potential
The electric potential at a point is the work done per unit positive charge in bringing a test charge from infinity to that point.
Potential Difference
The potential difference between two points A and B is:
Vₐ − V_B = W_BA / q
Where W_BA is the work done moving charge q from B to A.
- Unit: Volt (V)
- 1 Volt = 1 Joule / 1 Coulomb
The potential difference between two points is also called voltage.
Relation to Energy
Work done on a charge stores electric potential energy:
U = qV
How Potential Difference Causes Current to Flow
- A free positive charge naturally moves from higher to lower potential.
- A free negative charge (electron) moves from lower to higher potential.
- When a conducting wire connects two points at different potentials, electrons flow from lower potential to higher potential (conventional current flows opposite from higher to lower potential).
- Flow continues until both points reach equal potential then it stops.
Role of a Cell (Battery)
A cell maintains a constant potential difference by converting chemical energy into electrical energy. The terminal at higher potential is the positive terminal; the terminal at lower potential is the negative terminal.
A common dry cell provides 1.5 V. Two such cells in series provide 3 V.
A combination of cells is called a battery.
Electric Current
Definition
Electric current is the rate of flow of electric charge through a cross-section:
I = Q / t
Where:
- I = current (Amperes, A)
- Q = charge (Coulombs, C)
- t = time (seconds, s)
1 Ampere = 1 Coulomb per second
Direction of Current
By convention, the direction of conventional current is taken as the direction of flow of positive charges i.e., from the positive terminal to the negative terminal through the external circuit.
Electrons (negative charges) actually flow in the opposite direction from negative to positive terminal through the external circuit.
Concept of Resistance
In a metallic conductor, free electrons drift slowly under the influence of an applied potential difference. As they drift, they collide with atoms/ions of the conductor lattice these collisions impede the drift. This opposition to the flow of charge is called electrical resistance.
Facts about electron motion:
- Speed of electrons in random thermal motion: ~10⁵ m/s
- Drift speed of electrons under potential difference: ~10⁻⁴ m/s (very slow!)
- Speed at which current propagates: ~3 × 10⁸ m/s (speed of light) — this is why a bulb lights instantly even if the power source is far away.
Drift Velocity and Current
For a conductor of length l and cross-sectional area A, with n electrons per unit volume and drift velocity vd:
I = n × e × A × vd
Ohm's Law and Resistance
Ohm's Law
Statement: The electric current through a metallic conductor is directly proportional to the potential difference applied across its ends, provided the temperature remains constant.
V = IR
Or equivalently: I = V/R
Where R is the resistance of the conductor.
Resistance
Resistance is the property of a conductor to oppose the flow of electric charge through it.
R = V / I
Unit: Ohm (Ω)
Definition of 1 Ohm: The resistance of a conductor is 1 Ω if a potential difference of 1 V causes a current of 1 A to flow.
1 Ω = 1 V / 1 A
Factors Affecting Resistance
The resistance of a uniform conductor depends on:
- Length (l): R ∝ l (longer wire = more resistance)
- Cross-sectional area (A): R ∝ 1/A (thicker wire = less resistance)
- Material (ρ): R = ρ × l / A
Where ρ (rho) is the electrical resistivity (or specific resistance) of the material.
- SI unit of resistivity: Ω·m
- Good conductors have low resistivity; insulators have very high resistivity.
Series and Parallel Circuits
Series Connection of Resistors
In a series circuit, the same current flows through all resistors.
Total voltage: V = V₁ + V₂ + V₃
Equivalent resistance:
Rₑq = R₁ + R₂ + R₃ + ... + Rₙ
The equivalent resistance in series is always greater than the largest individual resistance.
Parallel Connection of Resistors
In a parallel circuit, the same potential difference is applied across all resistors.
Total current: I = I₁ + I₂ + I₃
Equivalent resistance:
1/Rₑq = 1/R₁ + 1/R₂ + 1/R₃ + ... + 1/Rₙ
For two resistors:
Rₑq = (R₁ × R₂) / (R₁ + R₂)
The equivalent resistance in parallel is always less than the smallest individual resistance.
Current Distribution in Parallel
Current through each branch is inversely proportional to its resistance:
I₁/I₂ = R₂/R₁
The branches carrying higher resistance receive less current.
Practical Comparison: Series vs. Parallel
| Feature | Series | Parallel |
|---|---|---|
| Current | Same through all | Divides among branches |
| Voltage | Divides among resistors | Same across all |
| Equivalent resistance | Greater than largest R | Less than smallest R |
| Effect of one failure | Entire circuit breaks | Other branches continue working |
| Application | Decorative fairy lights | Home wiring (lights, fans, appliances) |
Home appliances are connected in parallel so that each device operates at full mains voltage, and failure of one device does not affect others.
Kirchhoff's Laws (Advanced)
Kirchhoff's Current Law (KCL) – Junction Rule
The algebraic sum of currents at any junction is zero.
ΣI = 0 or I_in = I_out
Based on the Law of Conservation of Charge.
Kirchhoff's Voltage Law (KVL) – Loop Rule
The algebraic sum of potential changes around any closed loop is zero.
ΣΔV = 0 or Σξ = ΣIR
Based on the Law of Conservation of Energy.
Common Measuring Instruments
- Ammeter: Measures electric current always connected in series in a circuit.
- Voltmeter: Measures potential difference; always connected in parallel across the element being measured.
Heating Effect of Electric Current
11.1 Joule's Law of Heating
When current flows through a resistor, electrical energy is converted to heat. The heat produced is given by Joule's Law:
U = I² × R × t
Also expressed as:
U = V²t / R = VIt
Where:
- U = heat produced (Joules)
- I = current (Amperes)
- R = resistance (Ohms)
- t = time (seconds)
- V = potential difference (Volts)
Relationships
- For constant R and t: U ∝ I² → Doubling current → 4× the heat.
- For constant I and t: U ∝ R → More heat in higher resistance.
- For constant V and t: U ∝ 1/R → More heat in lower resistance.
Applications of the Heating Effect
| Appliance | Principle |
|---|---|
| Electric bulb | Tungsten filament heated to ~3000°C emits white light |
| Electric iron | Resistance heating element converts current to heat |
| Room heater | High-resistance coil radiates infrared heat |
| Toaster | Nichrome wire element toasts bread |
| Electric fuse | Thin wire of low melting point melts when current exceeds safe limit |
| Immersion heater | Heats water directly by immersed resistive element |
The Electric Bulb
- Filament material: Tungsten (melting point ~3400°C)
- Filament temperature during use: ~3000°C
- Bulb filling: Inert gas (argon or nitrogen) to prevent oxidation and maintain temperature
- Why inert gas? Prevents filament burning and slows evaporation of tungsten.
The Electric Fuse
A fuse is a safety device made of a thin metallic wire with a low melting point (typically tin, or tin-copper alloy), connected in series with the appliance.
How it works: If current exceeds the rated safe value, the fuse wire heats up rapidly and melts, breaking the circuit and preventing damage to appliances or wiring.
- Fuse for 5A circuits uses a thinner wire than fuse for 15A circuits.
- Modern circuits use circuit breakers (MCBs) which can be reset after tripping.
Disadvantages of Heating Effect
- Energy is wasted as heat in transmission lines.
- Excessive heat can damage electronic components (requires cooling fans, heat sinks, or ventilation grills in TVs, computers, and motors).
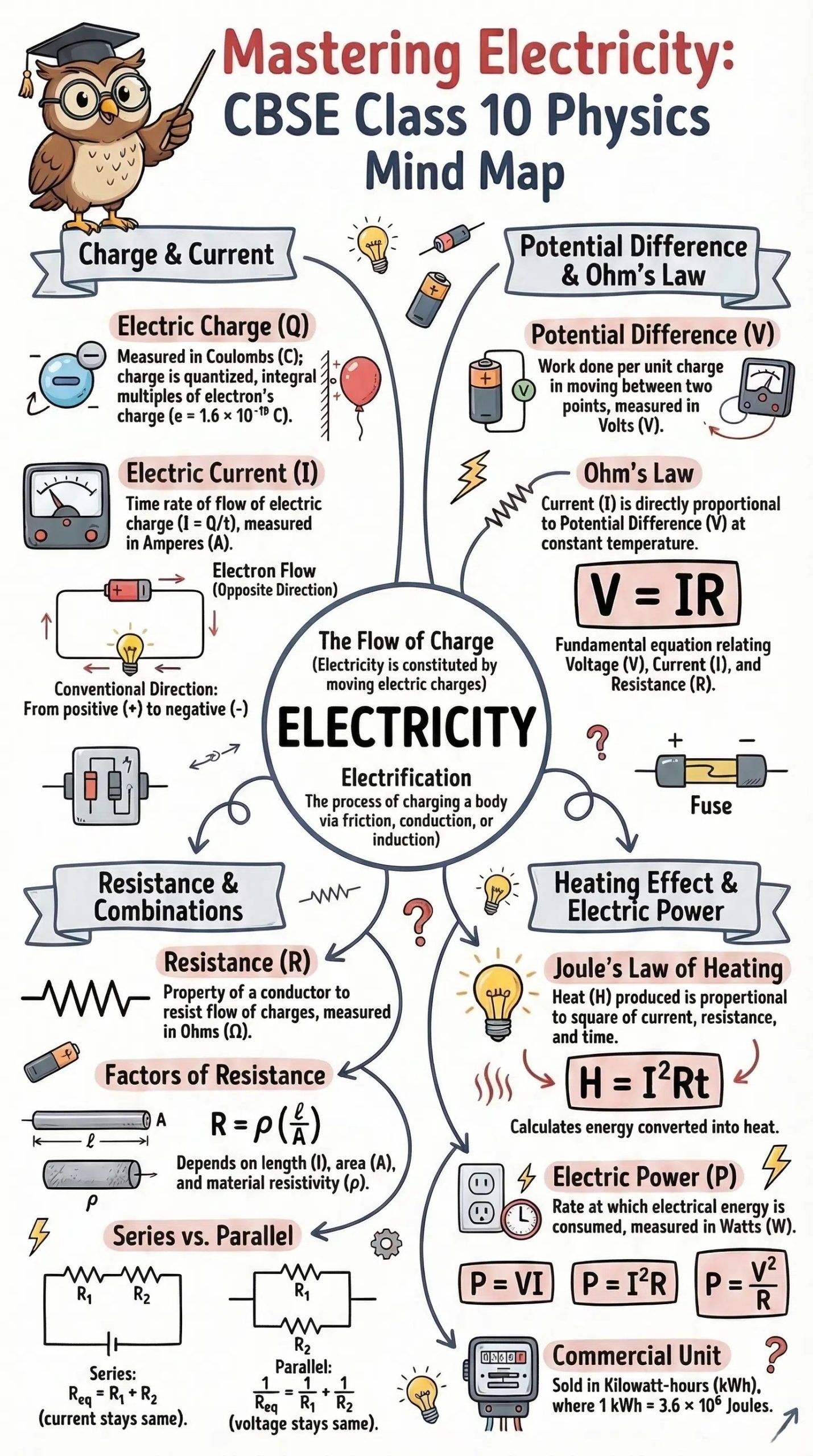
Electric Power and Commercial Units
Electric Power
Electric power is the electrical energy produced or consumed per unit time:
P = U / t = I²R = VI = V²/R
Unit: Watt (W)
1 Watt = 1 Joule/second
Rating of Electric Appliances
Every appliance is rated with power (W) and voltage (V), e.g., "60W, 220V."
This means:
- Apply 220V across the appliance → it consumes 60W.
- Its resistance: R = V²/P = (220)²/60 = 806.7 Ω
- Current drawn: I = P/V = 60/220 ≈ 0.27 A
Higher power rating = smaller resistance (for the same voltage rating).
Commercial Unit of Energy: kilowatt-hour (kWh)
1 kWh = 1000 W × 3600 s = 3.6 × 10⁶ J
1 kilowatt-hour = 1 unit of electricity
The cost of electricity is charged per kilowatt-hour (unit). This is what appears on your electricity bill.
Example: A 1000W heater used for 1 hour consumes 1 unit (1 kWh) of electricity.
Electricity Class 10 Physics Solved Examples
Q: To generate a charge of 1 μC on a sphere, how many electrons must be added?
Solution:
- 1 C requires 6.25 × 10¹⁸ electrons
- 1 μC = 10⁻⁶ C requires: 6.25 × 10¹⁸ × 10⁻⁶ = 6.25 × 10¹² electrons
Q: Two neutral bodies A and B, each having 6.25 × 10¹⁷ electrons added, are 1 m apart in air. Find the force.
Solution:
- Charge on each: q = 6.25 × 10¹⁷ / 6.25 × 10¹⁸ = 1/10 C = 0.1 C
- F = 9 × 10⁹ × (0.1 × 0.1) / 1² = 9 × 10⁹ × 10⁻² = 9 × 10⁷ N
Q: If charges are doubled and distance between them is doubled, what happens to force?
Solution:
- F₁ = k × q₁q₂/d²
- F₂ = k × (2q₁)(2q₂)/(2d)² = k × 4q₁q₂/4d² = k × q₁q₂/d² = F₁
- Force remains unchanged.
Q: Distance is quadrupled and each charge becomes 1/4 of original. Find ratio of final to initial force.
Solution:
- F₂/F₁ = [(1/4 × 1/4) / (4)²] = (1/16) / 16 = 1/256
- Final force = (1/256) × Initial force.
Q: Charges are doubled; force must remain same. By what factor must distance change?
Solution:
- k × q₁q₂/d² = k × (2q₁)(2q₂)/(xd)²
- 1 = 4/x² → x² = 4 → x = 2
- Distance must be doubled.
Q: Can an electron placed between +q and −q experience zero net force?
Solution: No. The +q attracts the electron leftward; the −q also repels (like charges... wait — electron is negative, −q repels electron leftward). Both forces act in the same direction. Net force cannot be zero at any position between A and B.
Q: Compare electrostatic force between two unit charges to gravitational force between two unit masses (1m apart).
Solution:
- Fɡ = 6.627 × 10⁻¹¹ N
- Fₑ = 9 × 10⁹ N
- Fₑ/Fɡ = 9 × 10⁹ / 6.627 × 10⁻¹¹ = 1.35 × 10²⁰
- Electrostatic force is 1.35 × 10²⁰ times greater.
Q: 6 × 10¹⁷ electrons cross a cross-section per minute. Find electric current.
Solution:
- Q = 6 × 10¹⁷ × 1.6 × 10⁻¹⁹ = 9.6 × 10⁻² C
- I = Q/t = 9.6 × 10⁻² / 60 = 1.6 × 10⁻³ A = 1.6 mA
Q: A 4V battery connected to a lamp of resistance 4Ω. Find current.
Solution:
- I = V/R = 4/4 = 1 A
Q: A 10Ω and 5Ω resistor connected in series. Find equivalent resistance.
Solution:
- Rₑq = 10 + 5 = 15 Ω
Q: 3Ω and 6Ω connected in parallel. Find equivalent resistance.
Solution:
- Rₑq = (3 × 6)/(3 + 6) = 18/9 = 2 Ω
Q: 1Ω connected in parallel with 100Ω. Find equivalent resistance.
Solution:
- Rₑq = (1 × 100)/(1 + 100) = 100/101 ≈ 0.99 Ω
- Note: Equivalent resistance is very close to the smaller resistance.
Q: Find resistance of a copper wire, length 1m, cross-section 2 mm², resistivity ρ = 1.7 × 10⁻⁸ Ω·m.
Solution:
- A = 2 mm² = 2 × 10⁻⁶ m²
- R = ρl/A = (1.7 × 10⁻⁸ × 1) / (2 × 10⁻⁶) = 8.5 × 10⁻³ Ω
Q: Wire of resistance R cut into 5 equal parts, connected in parallel. Find R/R'.
Solution:
- Each part = R/5
- 1/R' = 5 × (5/R) = 25/R → R' = R/25
- R/R' = 25
Q: A wire's diameter is doubled; length is same. How does resistance change?
Solution:
- R ∝ 1/d² (since A = πd²/4)
- New R₂ = R₁/4
- Resistance becomes one-fourth.
Q: From a V-I graph with V=4.8V corresponding to I=0.4A, find resistance.
Solution:
- R = V/I = 4.8/0.4 = 12 Ω
Q: 12V battery produces current of 2.5 mA. Find resistance.
Solution:
- R = V/I = 12 / (2.5 × 10⁻³) = 4800 Ω = 4.8 kΩ
Q: Three identical bulbs in parallel draw 6A total. One fuses. What is the new total current?
Solution:
- Each bulb draws 6/3 = 2A
- With one fused, 2 bulbs remain, each still drawing 2A
- New total = 2 + 2 = 4 A
Q: An electric bulb rated 220V, 100W is operated at 110V. Find power consumed.
Solution:
- R = V²/P = (220)²/100 = 484 Ω
- P' = V'²/R = (110)²/484 = 12100/484 = 25 W
Q: A 1000W heater runs for 1 hour. Calculate energy in kWh and joules, and the cost at ₹3/unit.
Solution:
- Energy = 1000W × 1h = 1 kWh = 1 unit
- In joules: 1 kWh = 3.6 × 10⁶ J
- Cost = 1 × ₹3 = ₹3
Class 10 Electricity Quick Reference Summary Table
| Quantity | Symbol | Formula | SI Unit |
|---|---|---|---|
| Electric Charge | Q | q = ne | Coulomb (C) |
| Electric Force | F | F = kq₁q₂/d² | Newton (N) |
| Electric Current | I | I = Q/t | Ampere (A) |
| Potential Difference | V | V = W/Q | Volt (V) |
| Resistance | R | R = V/I = ρl/A | Ohm (Ω) |
| Resistivity | ρ | ρ = RA/l | Ohm·metre (Ω·m) |
| Electric Power | P | P = VI = I²R = V²/R | Watt (W) |
| Heat Produced | U | U = I²Rt | Joule (J) |
| Energy (Commercial) | — | 1 kWh = 3.6 × 10⁶ J | kWh (Unit) |
Formulas at a Glance
- Coulomb's Law: F = 9 × 10⁹ × (q₁q₂)/d²
- Charge Quantisation: q = ±ne (e = 1.6 × 10⁻¹⁹ C)
- Electric Current: I = Q/t = nеAvd
- Ohm's Law: V = IR
- Series Resistance: Rₑq = R₁ + R₂ + R₃
- Parallel Resistance: 1/Rₑq = 1/R₁ + 1/R₂ + 1/R₃
- Resistivity: R = ρl/A
- Joule's Heating: U = I²Rt = V²t/R
- Electric Power: P = VI = I²R = V²/R
- Commercial Energy: 1 kWh = 3.6 × 10⁶ J
