Class 10 Chemical Reactions and Equations notes are the foundation of Chemistry for secondary school students. In class 10 science chapter 1 notes, students learn how substances change during a chemical reaction and how these changes are written using chemical equations.
This chapter explains important concepts such as reactants and products, balanced chemical equations, types of chemical reactions, corrosion, and rancidity. Understanding these topics helps students build strong basics for higher classes and competitive exams.
In these chemical reactions and equations class 10 notes ncert, we explain each concept with real-life examples. For example, burning of magnesium ribbon, rusting of iron, and digestion of food are common chemical changes we see around us.
The notes also include chemical reactions and equations class 10 notes questions answers to help students practice and revise important points.
Our aim is to make class 10 chemical reactions and equations easy to understand by breaking down complex topics into small, clear steps. Students can access for class 10 chemical reactions and equations notes pdf free download can use these notes for quick revision before exams.
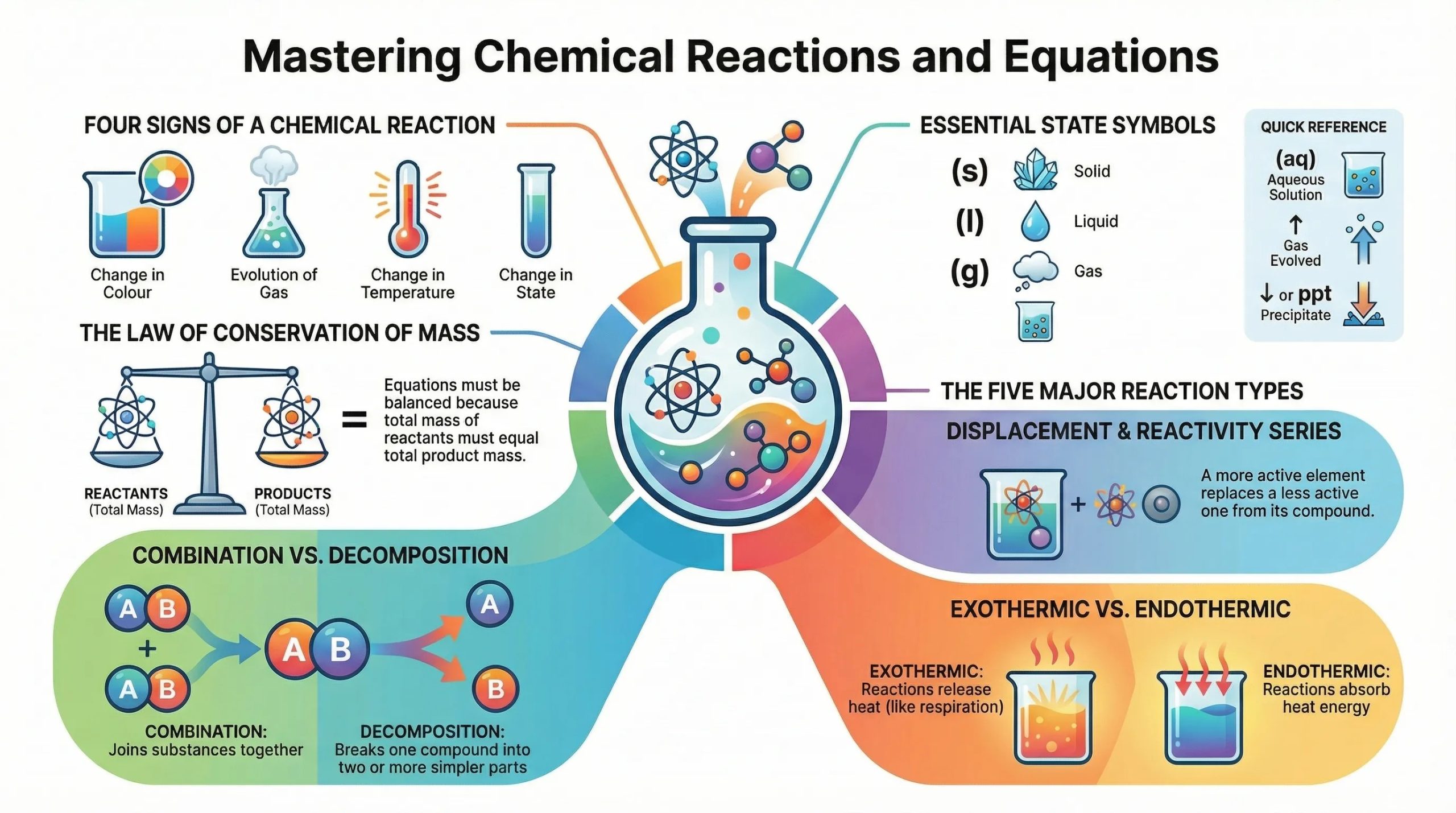
What is Chemical Reactions?
A chemical reaction is a process that transforms one or more substances into entirely new substances with different properties. The substances that participate in a reaction are called reactants, and the new substances formed are called products.
Example:
NaOH + HCl → NaCl + H₂O
Here, Sodium Hydroxide (NaOH) and Hydrochloric Acid (HCl) are reactants, while Sodium Chloride (NaCl) and Water (H₂O) are products.
At the atomic level, chemical reactions involve the breaking of bonds between atoms of reacting substances and the formation of new bonds in the products.
CBSE Class 10 Science Chemical Reactions and Equations Notes PDF Download
Fill the form to download this PDF
Indicators That a Chemical Reaction Has Taken Place
You can identify a chemical reaction by observing one or more of the following:
| Observation | Example |
|---|---|
| Change in state | Solid Mg + gaseous O₂ → solid MgO |
| Change in colour | Brown rust forming on black iron nails |
| Evolution of a gas | H₂ gas released when Zn reacts with HCl |
| Change in temperature | Flask warms up when Zn reacts with H₂SO₄ |
Physical vs. Chemical Changes
Understanding the difference between physical and chemical changes is foundational to chemistry.
Physical Changes
A change in which the physical properties of a substance are altered but the chemical composition remains unchanged.
Characteristics:
- Identity of the substance is maintained
- Change is reversible
- Heat change may or may not occur
- Only physical state or properties are altered
Examples: Freezing, melting, boiling, condensation
Chemical Changes
A change in which one or more substances are transformed into new substances with a different chemical composition.
Characteristics:
- Identity of the original substance is completely lost
- Change cannot be reversed
- Generally accompanied by an energy change
- Chemical composition is altered
Examples: Burning of a candle, rusting of iron, calcination of limestone
Chemical Equations
A chemical equation is a symbolic representation of a chemical reaction using symbols and formulae of reactants and products.
Types of Chemical Equations
(a) Word Equations
A word equation links the names of reactants with those of products.
Example:
Magnesium + Oxygen → Magnesium oxide
Rules for writing word equations:
- Reactants are written on the left-hand side (LHS), separated by +
- Products are written on the right-hand side (RHS), separated by +
- An arrow (→) points from reactants to products
(b) Symbol Equations
A brief representation using chemical symbols and formulae.
Example:
Mg + O₂ → MgO (unbalanced) 2Mg + O₂ → 2MgO (balanced)
Essentials of a Valid Chemical Equation
- It must represent a true chemical change
- All reactants and products must be written using their correct chemical formulae
- The total number of atoms of each element must be equal on both sides (Law of Conservation of Mass)
- It must be molecular in representation
Making Equations More Informative
To convey maximum information, chemical equations can include:
| Symbol | Meaning |
|---|---|
| (s) | Solid state |
| (l) | Liquid state |
| (g) | Gaseous state |
| (aq) | Aqueous solution |
| ↑ | Gas evolved |
| ↓ | Precipitate formed |
| + Heat | Exothermic reaction |
| – Heat | Endothermic reaction |
| ⇌ | Reversible reaction |
Example with state symbols:
2Na(s) + 2H₂O(l) → 2NaOH(aq) + H₂(g)↑
Balancing Chemical Equations
Why Balance?
An unbalanced (skeletal) equation does not satisfy the Law of Conservation of Mass, which states:
The total mass of all products in a chemical reaction equals the total mass of all reactants.
Hit and Trial Method (Steps)
- Write the word equation with reactants on the LHS and products on the RHS
- Convert to a symbol equation using correct formulae
- Count atoms of each element on both sides
- Adjust coefficients (not subscripts) to equalise atom counts
- Verify by re-counting all atoms
Example: Balancing Mg + HCl → MgCl₂ + H₂
| Element | LHS | RHS |
|---|---|---|
| Mg | 1 | 1 ✓ |
| Cl | 1 | 2 ✗ |
| H | 1 | 2 ✗ |
Place coefficient 2 before HCl:
Mg + 2HCl → MgCl₂ + H₂ ✓ (Balanced)
Limitations of Chemical Equations
A chemical equation does not directly convey:
- Physical state of reactants/products (unless state symbols are added)
- Conditions required (temperature, pressure, catalyst)
- Rate of the reaction
- Heat changes (unless specified)
- Concentration of reactants
- Mechanism of the reaction
- Whether the reaction is reversible or irreversible
Types of Chemical Reactions
(a) Combination Reactions
Two or more substances combine to form a single new substance.
General form: A + B → AB
Types:
- Two elements: C(s) + O₂(g) → CO₂(g)
- Element + compound: 2NO + O₂ → 2NO₂
- Two compounds: CaO(s) + H₂O(l) → Ca(OH)₂(s)(Quick lime + water → Slaked lime)
Note: The reaction of quick lime with water is highly exothermic and is used in whitewashing. The slaked lime then reacts with CO₂ in air to form calcium carbonate, giving walls their shine.
(b) Decomposition Reactions
A single compound breaks down into two or more simpler substances. These are the reverse of combination reactions.
General form: AB → A + B
Types of Decomposition:
i. Thermal Decomposition – Energy supplied as heat
2FeSO₄(s) →(Heat)→ Fe₂O₃(s) + SO₂(g) + SO₃(g)
CaCO₃(s) →(Heat)→ CaO(s) + CO₂(g)
ii. Electrolytic Decomposition (Electrolysis) – Energy supplied as electricity
2H₂O(l) →(Electric current)→ 2H₂(g) + O₂(g)
Note: In this reaction, the volume of hydrogen collected is double that of oxygen.
iii. Photolytic Decomposition – Energy supplied as light
2AgCl(s) →(Sunlight)→ 2Ag(s) + Cl₂(g)
This reaction forms the basis of black and white photography.
(c) Displacement Reactions
A more reactive element displaces a less reactive element from its compound.
Activity series (decreasing reactivity):
K > Na > Ba > Ca > Mg > Al > Zn > Fe > Ni > Sn > Pb > H > Cu > Hg > Ag > Au
Examples:
Fe(s) + CuSO₄(aq) → FeSO₄(aq) + Cu(s) (Blue copper sulphate solution turns green)
Zn(s) + CuSO₄(aq) → ZnSO₄(aq) + Cu(s)
(d) Double Displacement Reactions (Metathesis)
Two compounds react by exchanging ions to form new compounds.
General form: AB + CD → AD + CB
Types:
i. Precipitation Reactions – An insoluble precipitate is formed
Na₂SO₄(aq) + BaCl₂(aq) → 2NaCl(aq) + BaSO₄(s)↓ (White precipitate)
AgNO₃(aq) + NaCl(aq) → AgCl(s)↓ + NaNO₃(aq)
ii. Neutralisation Reactions – An acid reacts with a base to form salt and water
HCl(aq) + NaOH(aq) → NaCl(aq) + H₂O(l)
The net ionic equation is: H⁺(aq) + OH⁻(aq) → H₂O(l)
Application: Antacids work through neutralisation — they are simple bases that neutralise excess digestive acid in the stomach.
Oxidation and Reduction (Redox Reactions)
Classical Definitions
| Process | Classical Definition |
|---|---|
| Oxidation | Addition of oxygen OR removal of hydrogen |
| Reduction | Addition of hydrogen OR removal of oxygen |
| Oxidising agent | Substance that provides oxygen or removes hydrogen (gets reduced itself) |
| Reducing agent | Substance that provides hydrogen or removes oxygen (gets oxidised itself) |
Rule: Oxidation and reduction always occur simultaneously — hence the term redox reaction.
Worked Example: CuO + H₂ → Cu + H₂O
Oxidation (H2 gains oxygen)
↓
CuO + H2 → (Heat) → Cu + H2O
↓
Reduction (CuO loses oxygen)
- H₂ is oxidised (gains oxygen) → H₂ is the reducing agent
- CuO is reduced (loses oxygen) → CuO is the oxidising agent
Electronic Concept of Oxidation and Reduction
For ionic reactions:
| Process | Electronic Definition |
|---|---|
| Oxidation | Loss of electrons |
| Reduction | Gain of electrons |
| Oxidising agent | Accepts electrons; gets reduced |
| Reducing agent | Donates electrons; gets oxidised |
Examples:
- Mg → Mg²⁺ + 2e⁻ (Oxidation)
- Zn²⁺ + 2e⁻ → Zn (Reduction)
Oxidation Number and Valency
Valency
The combining capacity of an element always a positive whole number with no sign. Noble gases have zero valency.
Oxidation Number (Oxidation State)
A positive or negative number assigned to an atom in a compound based on prescribed rules. Can be fractional or zero.
Rules for Assigning Oxidation Numbers
- Free element → Oxidation number = 0 (e.g., F₂, P₄, Ag)
- Monoatomic ion → Oxidation number = charge on the ion (e.g., Ca²⁺ = +2)
- Oxygen = –2 (except in peroxides where it is –1; in OF₂ it is +2)
- Hydrogen = +1 (except in metallic hydrides where it is –1)
- Group I metals = +1; Group II metals = +2
- Halogens in binary ionic compounds = –1
- Sum of oxidation numbers in a compound = 0
- Sum of oxidation numbers in a polyatomic ion = charge on the ion
Relationship: Oxidation Number vs. Reaction Type
| Process | Effect on Oxidation Number |
|---|---|
| Oxidation | Oxidation number increases |
| Reduction | Oxidation number decreases |
| Oxidising agent undergoes | Oxidation number decreases |
| Reducing agent undergoes | Oxidation number increases |
Sample Calculation: Oxidation Number of Mn in KMnO₄
Let oxidation number of Mn = x
+1 + x + 4(–2) = 0 x = +7
Balancing Redox Reactions
Method 1: Oxidation Number Method
Steps:
- Write the skeleton equation
- Assign oxidation numbers to all atoms
- Identify atoms that change oxidation number
- Calculate total increase and decrease in oxidation number
- Multiply formulae so that total increase = total decrease
- Balance remaining atoms (excluding H and O first)
- Finally balance H and O atoms
Method 2: Ion-Electron (Half-Reaction) Method
Steps:
- Write the ionic equation
- Split into oxidation and reduction half-reactions
- Balance each half-reaction for atoms and charge
- In acidic medium: add H₂O to balance O; add H⁺ to balance H
- In basic medium: add OH⁻ to balance charge; add H₂O to balance OH⁻
- Multiply half-reactions so electrons cancel
- Add the two half-reactions and cancel common terms
Effects of Oxidation Reactions in Daily Life
Respiration
Respiration is an exothermic oxidation reaction occurring in body cells:
C₆H₁₂O₆(aq) + 6O₂(aq) → 6CO₂(aq) + 6H₂O(l) + Energy
This energy keeps the heart, muscles, and all vital functions working.
Combustion Reactions
Any reaction in which a substance burns in the presence of oxygen:
CH₄(g) + 2O₂(g) → CO₂(g) + 2H₂O(l) (Methane/Natural gas)
C₄H₁₀(g) + 13/2 O₂(g) → 4CO₂(g) + 5H₂O(l) (Butane/LPG)
Environmental Note: Combustion releases CO, SO₂, SO₃, and NO₂ into the atmosphere, contributing to air pollution.
Corrosion and Rancidity
Corrosion
Definition: The gradual eating up of metals by the action of air, moisture, or chemicals (such as acids) on their surface.
Rusting of Iron is the most common form of corrosion:
2Fe + 3/2 O₂ + xH₂O → Fe₂O₃·xH₂O (Hydrated ferric oxide / Rust)
Rust is a soft, porous, red-brown substance that continuously falls off, exposing fresh metal underneath — making rusting a self-perpetuating process.
Other examples:
- Black coating on silver (Ag₂S)
- Green coating on copper (basic copper carbonate)
Useful Corrosion: Aluminium forms a protective layer of Al₂O₃ on its surface that prevents further corrosion, making aluminium foils safe for food packaging.
Rancidity
Definition: The condition produced by aerial oxidation of fats and oils in food, marked by unpleasant smell and taste.
Rancidity makes food stale, unfit for eating, and sometimes toxic.
Prevention of Rancidity
| Method | Mechanism |
|---|---|
| Adding antioxidants (BHA, BHT) | Chemically prevent oxidation |
| Packaging in nitrogen gas | Eliminates oxygen contact |
| Refrigeration | Low temperature slows oxidation |
| Air-tight containers | Reduces oxygen exposure |
| Storing away from light | Prevents light-catalysed oxidation |
Natural antioxidants: Vitamin E and Vitamin C (ascorbic acid) occur naturally in fats and serve as antioxidants.
Solved Illustrations
Illustration 1: Balance the Given Equations
(i) CH₄ + O₂ → CO₂ + H₂O (unbalanced)
Solution: Count atoms: C=1, H=4 vs 2✗, O=2 vs 3✗
Place coefficient 2 before H₂O: CH₄ + O₂ → CO₂ + 2H₂O Now O: 2 vs 4 — place 2 before O₂:
CH₄ + 2O₂ → CO₂ + 2H₂O
(ii) Fe + O₂ → Fe₂O₃ (unbalanced)
Solution: Fe needs 2, O₂ needs 3 to balance (LCM of 2 and 3 = 6 Fe atoms, 4 Fe₂O₃):
4Fe + 3O₂ → 2Fe₂O₃
Illustration 2: Why Must a Chemical Equation Be Balanced?
Question: Why is it necessary to balance a chemical equation?
Solution: To satisfy the Law of Conservation of Mass, the number of atoms of every element must be equal on both sides of a chemical equation. An unbalanced (skeletal) equation does not fulfil this requirement. Balancing ensures that the total mass of reactants equals the total mass of products.
Illustration 3: Identify the Type of Reaction
(a) 2KNO₃ → 2KNO₂ + O₂ → Decomposition reaction (one compound breaking into two)
(b) N₂ + 3H₂ → 2NH₃ → Combination reaction (two elements combining into one compound)
(c) CuSO₄ + Fe → FeSO₄ + Cu → Displacement reaction (more reactive Fe displaces less reactive Cu)
(d) AgNO₃(aq) + NaCl(aq) → AgCl(s)↓ + NaNO₃(aq) → Double displacement (precipitation) reaction
Illustration 4: Conditions Required for Decomposition Reactions
Question: What conditions are required for decomposition reactions?
Solution: Decomposition reactions require energy input in the form of:
- Heat (thermal decomposition) — e.g., CaCO₃ → CaO + CO₂
- Light (photolytic decomposition) — e.g., 2AgCl → 2Ag + Cl₂
- Electricity (electrolytic decomposition) — e.g., 2H₂O → 2H₂ + O₂
Illustration 5: Balance by Oxidation Number Method
Equation: CuO + NH₃ → Cu + N₂ + H₂O
Solution:
Step 1 — Assign oxidation numbers:
- Cu in CuO: +2 → Cu: 0 (decrease of 2 — reduction)
- N in NH₃: –3 → N in N₂: 0 (increase of 3 — oxidation)
Step 2 — Equalise changes:
- To balance: 3 CuO provides decrease of 6; 2 NH₃ provides increase of 6
Step 3 — Balance remaining atoms: 3CuO + 2NH₃ → 3Cu + N₂ + 3H₂O
Balanced equation: 3CuO + 2NH₃ → 3Cu + N₂ + 3H₂O
Illustration 6: Balance by Ion-Electron Method
Equation: Cr₂O₇²⁻ + Fe²⁺ + H⁺ → Cr³⁺ + Fe³⁺ + H₂O
Solution:
Oxidation half-reaction: Fe²⁺ → Fe³⁺ + e⁻ ... (i)
Reduction half-reaction: Cr₂O₇²⁻ + 6e⁻ + 14H⁺ → 2Cr³⁺ + 7H₂O ... (ii)
Multiply (i) × 6 and add to (ii):
6Fe²⁺ + Cr₂O₇²⁻ + 14H⁺ → 6Fe³⁺ + 2Cr³⁺ + 7H₂O ✓
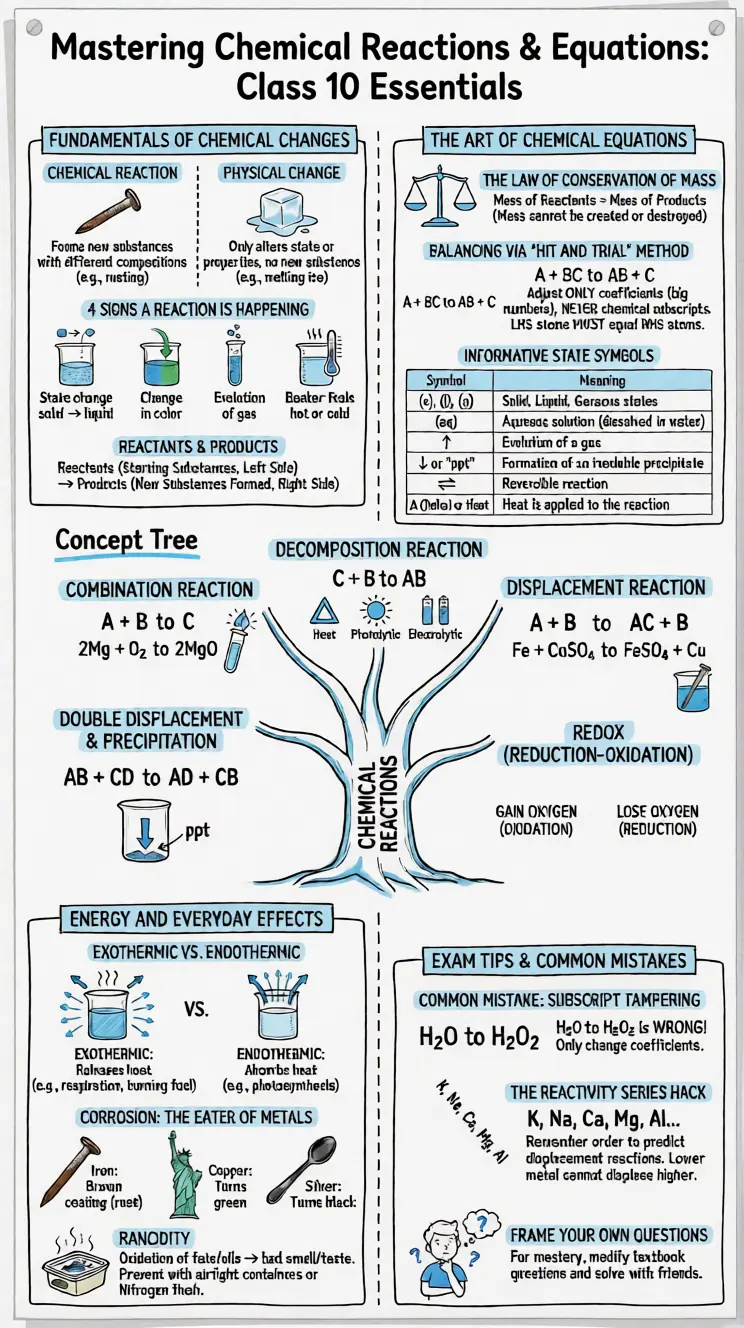
Points to Remember
- A chemical reaction involves breaking of old bonds and formation of new bonds.
- Indicators of chemical reactions: change in state, colour, evolution of gas, or temperature change.
- A balanced equation satisfies the Law of Conservation of Mass.
- Combination: A + B → AB (multiple reactants, single product)
- Decomposition: AB → A + B (single reactant, multiple products; reverse of combination)
- Displacement: A + BC → AC + B (more reactive element displaces less reactive)
- Double displacement: AB + CD → AD + CB (exchange of ions)
- Oxidation = gain of oxygen / loss of hydrogen / loss of electrons / increase in oxidation number
- Reduction = loss of oxygen / gain of hydrogen / gain of electrons / decrease in oxidation number
- Oxidation and reduction occur simultaneously in a redox reaction.
- Corrosion = gradual destruction of metals by air/moisture; rusting of iron forms Fe₂O₃·xH₂O.
- Rancidity = oxidation of fats/oils in food, causing bad smell/taste; prevented by antioxidants, refrigeration, nitrogen packaging, or airtight storage.
- Valency is always a positive whole number; oxidation number can be negative, fractional, or zero.
