Class 10 Science Chapter 4 Carbon And Its Compounds: Class 10 Science Chapter 4 Carbon And Its Compounds is one of the most important chapters in the CBSE syllabus. In this chapter, students study about carbon and its compounds class 10, their properties, structure, and uses in daily life. Carbon is a unique element because it forms a large number of compounds due to its tetravalency and catenation property. These special properties allow carbon to form covalent bonds and long chains, which are the basis of organic chemistry.
In carbon and its compounds class 10 notes, students learn about covalent bonding, electron sharing, hydrocarbons, homologous series, and functional groups. The chapter also explains important topics like chemical properties of carbon compounds, combustion, oxidation, addition reactions, and substitution reactions. Concepts such as ethanol, ethanoic acid, soaps and detergents are also discussed with real-life examples.
Students often search for carbon and its compounds class 10 notes ncert and class 10 carbon and its compounds notes pdf to revise important definitions, chemical equations, and diagrams. These resources help in better exam preparation and concept clarity. Understanding carbon and its compounds class 10 is very essential because it builds the base for higher studies in chemistry, although some students may find bonding concept little confusing at first but regular practice makes it easier.

What is Carbon and Its Compounds?
Carbon is one of the most remarkable elements in the periodic table. Although it constitutes only 0.02% of the Earth's crust (in the form of minerals like carbonates, hydrogen carbonates, coal, and petroleum) and just 0.03% of the atmosphere (as carbon dioxide), its importance is immeasurable.
Carbon forms the backbone of all known life on Earth every living organism, from bacteria to blue whales, is built on carbon-based chemistry. Beyond biology, carbon compounds are found in the clothes we wear, the medicines we take, the food we eat, fertilisers, fuels, plastics, and countless other everyday materials.
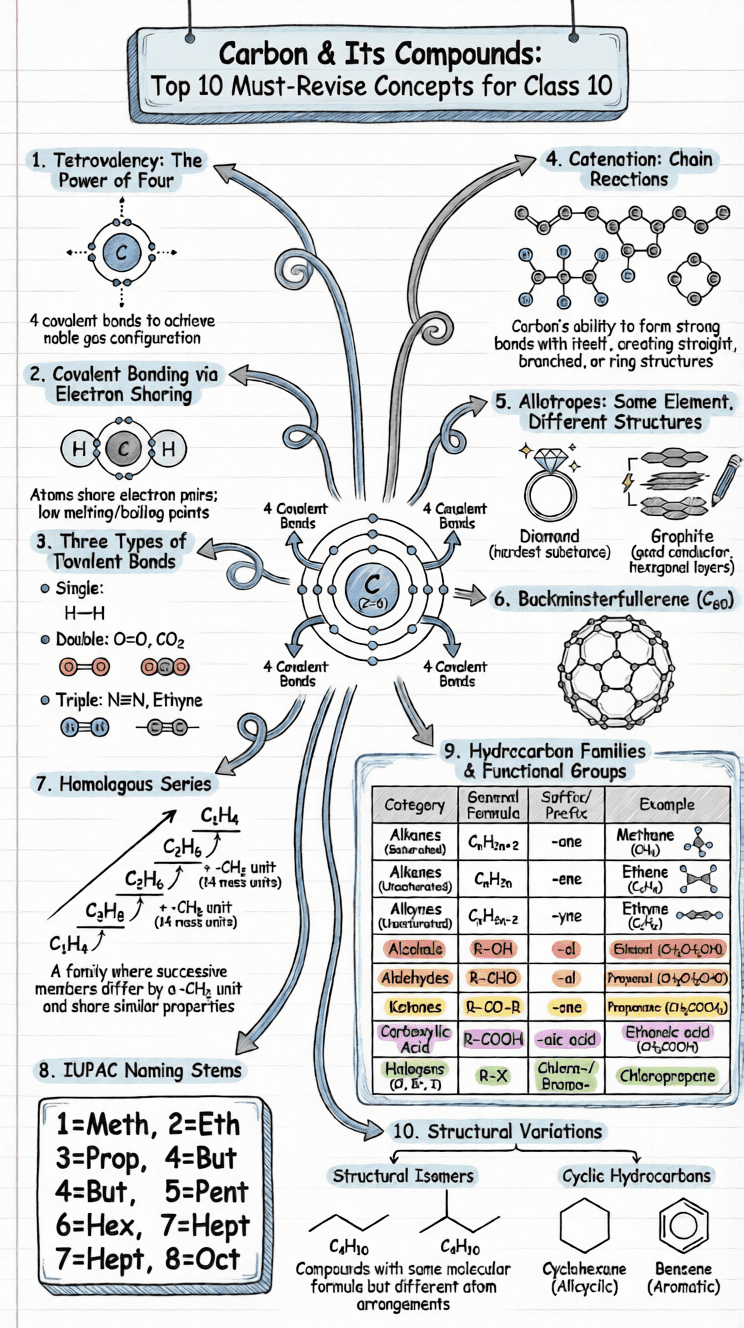
What makes carbon so exceptional is the sheer number and diversity of compounds it can form. More than three million carbon compounds are known — a number greater than the combined total of compounds formed by all other elements. This remarkable diversity arises from two key properties: tetravalency and catenation.
Class 10 Science Chapter 4 Carbon and Its Compounds Revison Notes PDF
Fill the form to download this PDF
Bonding in Carbon – Covalent Bond
Why Does Carbon Form Only Covalent Bonds?
Carbon has an atomic number of 6, with the electronic configuration:
- Shell K: 2 electrons
- Shell L (Outermost): 4 electrons
To achieve the stable noble gas configuration (like Neon, with 8 electrons in the outer shell), carbon would need to either gain 4 electrons or lose 4 electrons. Both these pathways require enormous amounts of energy and are not feasible under normal conditions. Instead, carbon achieves stability by sharing its 4 valence electrons with other atoms, forming covalent bonds.
Definition of Covalent Bond
A covalent bond is a chemical bond formed between two atoms of the same or different elements by the mutual sharing of one or more pairs of electrons, such that each atom attains the electronic configuration of the nearest noble gas.
Conditions for Covalent Bond Formation
- The combining atoms must have non-metallic character.
- The atoms should have 1 to 7 electrons in their valence shells (hydrogen, with just 1 valence electron, is a notable exception that still forms covalent bonds to complete its duplet).
- Each atom contributes an equal number of electrons to the shared pair.
- After sharing, each atom attains a stable electronic configuration.
Classification of Covalent Bonds
Based on the number of electron pairs shared, covalent bonds are of three types:
Single Covalent Bond (–)
Formed by the sharing of one pair of electrons.
Examples: H–H (hydrogen), Cl–Cl (chlorine), H–Cl (hydrochloric acid), CH₃–CH₃ (ethane)
Double Covalent Bond (=)
Formed by the sharing of two pairs of electrons.
Examples: O=O (oxygen), O=C=O (carbon dioxide), H₂C=CH₂ (ethene/ethylene)
Triple Covalent Bond (≡)
Formed by the sharing of three pairs of electrons.
Examples: N≡N (nitrogen), H–C≡C–H (ethyne/acetylene)
Important Point: As the number of shared pairs increases (single → double → triple), bond strength increases and bond length decreases.
Formation of Covalent Compounds – Examples
Understanding how covalent compounds form is central to this chapter. Here are the most important examples:
| Compound | Formula | Bond Type | How It Forms |
|---|---|---|---|
| Hydrogen | H₂ | Single | Each H atom shares its 1 electron; both complete their duplet (2 electrons) |
| Chlorine | Cl₂ | Single | Each Cl atom (configuration 2,8,7) shares 1 electron to complete its octet |
| Hydrochloric Acid | HCl | Single | H shares 1e⁻ with Cl; both achieve stable configurations |
| Oxygen | O₂ | Double | Each O atom (configuration 2,6) shares 2 electrons to complete its octet |
| Nitrogen | N₂ | Triple | Each N atom (configuration 2,5) shares 3 electrons to complete its octet |
| Water | H₂O | Single (×2) | O shares 1 electron each with two H atoms |
| Carbon Dioxide | CO₂ | Double (×2) | C (2,4) forms two double bonds with two O atoms |
| Methane | CH₄ | Single (×4) | C shares 1 electron each with four H atoms |
| Ethene | C₂H₄ | Double (C=C) | Two C atoms share 2 pairs; each C also bonds to 2 H atoms |
| Ethyne | C₂H₂ | Triple (C≡C) | Two C atoms share 3 pairs; each C also bonds to 1 H atom |
Polar and Non-Polar Covalent Bonds
Non-Polar Covalent Bond
When a covalent bond forms between two atoms of the same element (or atoms of equal electronegativity), the shared electron pair is located exactly midway between the two atoms. There is no charge separation.
Examples: H₂, O₂, N₂, Cl₂
Polar Covalent Bond
When a covalent bond forms between atoms of two different elements with different electronegativities, the shared electron pair is drawn closer to the more electronegative atom, creating a partial negative charge (δ⁻) on that atom and a partial positive charge (δ⁺) on the other.
Examples: HCl, H₂O, NH₃
In HCl, chlorine is more electronegative than hydrogen. The shared electrons lie closer to Cl, making Cl partially negative (δ⁻) and H partially positive (δ⁺).
Characteristics of Covalent Compounds vs Ionic Compounds
| Property | Ionic Compounds | Covalent Compounds |
|---|---|---|
| Bond formation | Transfer of electrons | Sharing of electrons |
| Physical state | Usually crystalline solids | Usually gases or liquids; few solids |
| Melting/Boiling point | High | Low (exceptions: diamond, graphite) |
| Solubility | Soluble in water; insoluble in organic solvents | Soluble in organic solvents; mostly insoluble in water |
| Electrical conductivity | Conduct electricity (in solution/molten state) | Usually non-conductors |
| Ionisation in water | Highly polar; ionise easily (e.g., NaCl → Na⁺ + Cl⁻) | Usually non-polar; do not ionise (e.g., glucose) |
Organic Compounds and Allotropes of Carbon
Organic Compounds
Organic compounds are those found in or derived from living organisms. Historically, scientists believed that a mysterious "Vital Force" was required to produce them until Friedrich Wöhler (1828) synthesised urea (an organic compound) in his laboratory from inorganic ammonium cyanate, shattering the Vital Force theory.
Allotropy and Allotropes of Carbon
Allotropy is the phenomenon where an element exists in two or more physically distinct forms (allotropes) that have similar chemical properties but different physical properties. Carbon has three major allotropes:
Diamond
- Structure: Each carbon atom is covalently bonded to four other carbon atoms in a tetrahedral arrangement, forming a rigid 3D network.
- Properties: Hardest natural substance; very high melting point; transparent and colourless; non-conductor of heat and electricity; refractive index of 2.45; density 3.51 g/cm³.
- Uses: Cutting tools (glass, marble), surgical instruments, drill bits, precision thermometers, spacecraft windows.
Graphite
- Structure: Carbon atoms are arranged in hexagonal layers. Within each layer, each carbon bonds to three others (leaving one free electron per atom). Layers are held together by weak van der Waals forces.
- Properties: Greyish-black, soft, slippery (soapy touch), good conductor of electricity (due to free electrons); density 2.26 g/cm³; very high melting point.
- Uses: Electrodes in dry cells and electric arc furnaces; dry lubricant; lead pencil cores; neutron moderator in nuclear reactors; manufacture of artificial diamonds.
Fullerene (C₆₀)
- Discovered in 1985 by Robert F. Curl Jr., Harold Kroto, and Richard E. Smalley (Nobel Prize in Chemistry, 1996).
- Buckminsterfullerene (C₆₀): Named after American architect R. Buckminster Fuller. It is nearly spherical, consisting of 12 pentagonal and 20 hexagonal faces with 60 corners resembling a football.
- Properties: Soluble in benzene (deep violet solution), crystalline form has semiconductor properties; fullerides (compounds with alkali metals) are superconductors.
- Uses: Superconductors, semiconductors, lubricants, catalysts, fibre reinforcement in plastics.
Versatile Nature of Carbon
The enormous number of carbon compounds (~3 million) is attributed to four key characteristics:
(1) Catenation
Catenation is the property of an element to form bonds with its own atoms, creating chains of varying lengths.
Carbon has the maximum catenation tendency of all elements, due to the exceptional strength of C–C bonds. Carbon chains can be:
- Straight chains: –C–C–C–
- Branched chains: Carbon chain with side branches
- Closed chains (rings): Carbon atoms forming cyclic structures (minimum 3 carbon atoms needed)
(2) Tetravalency
With 4 valence electrons, carbon forms four covalent bonds simultaneously with other carbon atoms, hydrogen, oxygen, nitrogen, sulphur, or halogens. This tetravalency allows for enormous structural diversity.
(3) Tendency to Form Multiple Bonds
Due to its small atomic size, carbon readily forms double bonds (C=C, C=O) and triple bonds (C≡C, C≡N). Multiple bonds increase the complexity of molecular architecture.
(4) Isomerism
Compounds with the same molecular formula but different structural arrangements are called isomers, and the phenomenon is called isomerism.
Example: C₂H₆O has two isomers:
- Ethanol (C₂H₅OH) — an alcohol
- Dimethyl ether (CH₃–O–CH₃) — an ether
Example: C₅H₁₂ (pentane) has three isomers:
- n-Pentane (straight chain)
- Iso-pentane (one branch)
- Neo-pentane (two branches)
Hydrocarbons - Saturated and Unsaturated
Hydrocarbons are compounds containing only carbon and hydrogen.
Classification of Hydrocarbons
- Acyclic (Open Chain)
- Saturated - Alkanes (CnH2n+2) - Single bonds only
- Unsaturated
- Alkenes (CnH2n) - Contains C=C double bond
- Alkynes (CnH2n−2) - Contains C≡C triple bond
- Cyclic (Closed Chain)
- Alicyclic - Carbon ring (Cyclopropane, Cyclobutane, etc.)
- Aromatic - Benzene ring structure
Alkanes (Saturated Hydrocarbons) – General Formula: CₙH₂ₙ₊₂
| No. of C atoms | Name | Formula |
|---|---|---|
| 1 | Methane | CH₄ |
| 2 | Ethane | C₂H₆ |
| 3 | Propane | C₃H₈ |
| 4 | Butane | C₄H₁₀ |
| 5 | Pentane | C₅H₁₂ |
| 6 | Hexane | C₆H₁₄ |
Alkenes (Unsaturated – Double Bond) – General Formula: CₙH₂ₙ
| No. of C atoms | Name | Formula |
|---|---|---|
| 2 | Ethene (Ethylene) | C₂H₄ |
| 3 | Propene (Propylene) | C₃H₆ |
| 4 | Butene (Butylene) | C₄H₈ |
Alkynes (Unsaturated – Triple Bond) – General Formula: CₙH₂ₙ₋₂
| No. of C atoms | Name | Formula |
|---|---|---|
| 2 | Ethyne (Acetylene) | C₂H₂ |
| 3 | Propyne (Methyl acetylene) | C₃H₄ |
| 4 | Butyne (Dimethyl acetylene) | C₄H₆ |
Chains, Branches, and Rings
Alicyclic Hydrocarbons
These contain rings of three or more carbon atoms and are named by adding the prefix "cyclo" to the corresponding straight-chain hydrocarbon name:
- Cyclopropane (C₃H₆), Cyclobutane (C₄H₈), Cyclopentane (C₅H₁₀), Cyclohexane (C₆H₁₂)
Aromatic Hydrocarbons
These contain at least one benzene ring a six-carbon ring with three alternating double bonds. The benzene ring is often represented as a hexagon with a circle inside to denote delocalised electrons.
Functional Groups
A functional group is an atom or group of atoms in an organic molecule that is primarily responsible for the compound's characteristic chemical properties.
Different compounds sharing the same functional group belong to the same chemical family and exhibit similar chemical properties.
Important Functional Groups in Carbon Compounds
| Heteroatom | Functional Group | Formula | Family Name |
|---|---|---|---|
| Halogen (F, Cl, Br, I) | Halo | –F, –Cl, –Br, –I | Haloalkanes |
| Oxygen | Alcohol/Hydroxyl | –OH | Alcohols |
| Oxygen | Aldehyde | –CHO | Aldehydes |
| Oxygen | Ketone | –CO– | Ketones |
| Oxygen | Carboxylic Acid | –COOH | Carboxylic Acids |
| Nitrogen | Nitro | –NO₂ | Nitro compounds |
| Nitrogen | Amine | –NH₂ | Amines |
Homologous Series
A homologous series is a group of organic compounds that have the same functional group, similar chemical properties, the same general formula, and in which successive members differ by a –CH₂ group (molecular mass difference of 14 u).
Characteristics of a Homologous Series
- All members share a common general formula (e.g., CₙH₂ₙ₊₂ for alkanes).
- Each successive member differs by –CH₂ (14 mass units).
- All members show similar chemical properties.
- All members can be prepared by similar methods.
- Physical properties (melting point, boiling point, density) change gradually with increasing molecular mass:
- C₁–C₄: Gases
- C₅–C₁₃: Liquids
- C₁₄ and above: Solids
Homologous Series Table
| Alkane (CₙH₂ₙ₊₂) | Alkene (CₙH₂ₙ) | Alkyne (CₙH₂ₙ₋₂) |
|---|---|---|
| Methane (CH₄) | – | – |
| Ethane (C₂H₆) | Ethene (C₂H₄) | Ethyne (C₂H₂) |
| Propane (C₃H₈) | Propene (C₃H₆) | Propyne (C₃H₄) |
| Butane (C₄H₁₀) | Butene (C₄H₈) | Butyne (C₄H₆) |
| Pentane (C₅H₁₂) | Pentene (C₅H₁₀) | Pentyne (C₅H₈) |
Nomenclature of Carbon Compounds (IUPAC)
The International Union of Pure and Applied Chemistry (IUPAC) has devised a systematic, universally accepted method for naming organic compounds.
Steps for Naming
- Identify the longest continuous chain of carbon atoms this gives the parent hydrocarbon name.
- Identify the functional group it is expressed using a suffix (or prefix).
- Number the carbon chain from the end closer to the principal functional group.
IUPAC Stems for Carbon Chain Lengths
| No. of C atoms | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 |
|---|---|---|---|---|---|---|---|---|---|---|
| Stem | Meth | Eth | Prop | But | Pent | Hex | Hept | Oct | Non | Dec |
Suffix Guide
| Bond/Functional Group | Suffix | Example |
|---|---|---|
| Single bond (Alkane) | -ane | Propane |
| Double bond (Alkene) | -ene | Propene |
| Triple bond (Alkyne) | -yne | Propyne |
| Alcohol (–OH) | -ol | Propanol |
| Aldehyde (–CHO) | -al | Propanal |
| Ketone (–CO–) | -one | Propanone |
| Carboxylic acid (–COOH) | -oic acid | Propanoic acid |
Prefix Guide (for substituents)
| Substituent | Prefix |
|---|---|
| –Cl | Chloro |
| –Br | Bromo |
| –I | Iodo |
Chemical Properties of Carbon Compounds
(A) Combustion
Carbon compounds burn in oxygen to produce carbon dioxide, water vapour, heat, and light. This is an exothermic oxidation reaction.
Complete Combustion (excess oxygen):
CH₄ + 2O₂ → CO₂ + 2H₂O + Heat + Light (Blue flame)
Incomplete Combustion (limited oxygen):
2CH₄ + 3O₂ → 2CO + 4H₂O (Smoky/yellow flame)
Nature of Flame:
- Saturated hydrocarbons (alkanes, LPG, natural gas) burn with a clean blue flame when oxygen is sufficient.
- Unsaturated hydrocarbons (alkenes, alkynes) burn with a luminous yellow/smoky flame due to higher carbon content.
- Alcohol and acetone undergo complete combustion, producing non-luminous flames with no carbon deposits.
LPG (Butane) Combustion:
C₄H₁₀ + 13/2 O₂ → 4CO₂ + 5H₂O + Heat + Light
Alcohol (Ethanol) Combustion:
C₂H₅OH + 3O₂ → 2CO₂ + 3H₂O + Heat + Light
Coal and Petroleum
Both coal and petroleum are fossil fuels formed from the remains of ancient organisms over millions of years under heat and pressure. Combustion of coal and petroleum releases sulphur and nitrogen oxides (SO₂, NO, NO₂), which are major environmental pollutants.
(B) Oxidation
Carbon compounds can be oxidised to different products depending on the oxidising agent used.
Ethanol Oxidation:
With chromic anhydride (CrO₃) in acetic acid:
CH₃CH₂OH + [O] → CH₃CHO + H₂O (Ethanol → Ethanal/Aldehyde)
With alkaline KMnO₄ (or acidified K₂Cr₂O₇) + heat:
CH₃CH₂OH + 2[O] → CH₃COOH + H₂O (Ethanol → Ethanoic Acid)
Experiment Note: When alkaline KMnO₄ is added drop by drop to ethanol, the purple colour initially discharges as ethanol is oxidised. Once all ethanol is converted, excess KMnO₄ retains its purple colour — confirming complete oxidation to ethanoic acid.
(C) Addition Reactions
Unsaturated hydrocarbons (alkenes and alkynes) undergo addition reactions, where atoms of another molecule are added across the double or triple bond to form a saturated compound.
Hydrogenation of Ethene:
C₂H₄ + H₂ → C₂H₆ (Ni catalyst, 250°C)
Hydrogenation of Ethyne (two steps):
C₂H₂ + H₂ → C₂H₄ → C₂H₄ + H₂ → C₂H₆ (Ni catalyst, heat)
Hydrogenation of Vegetable Oils: Unsaturated vegetable oils (groundnut oil, mustard oil) contain C=C double bonds. In the presence of nickel (Ni) catalyst and heat, these are hydrogenated to form vanaspati ghee, a semi-solid saturated fat.
Vegetable oil (unsaturated) + H₂ →(Ni, Heat) Vanaspati Ghee (saturated)
(D) Substitution Reactions
In substitution reactions, one or more hydrogen atoms in a saturated hydrocarbon are replaced by other atoms (typically halogens).
Chlorination of Methane (Photochemical reaction):
CH₄ + Cl₂ →(Sunlight) CH₃Cl + HCl (Chloromethane)
CH₃Cl + Cl₂ →(Sunlight) CH₂Cl₂ + HCl (Dichloromethane)
CH₂Cl₂ + Cl₂ →(Sunlight) CHCl₃ + HCl (Trichloromethane/Chloroform)
CHCl₃ + Cl₂ →(Sunlight) CCl₄ + HCl (Carbon Tetrachloride)
Note: Substitution reactions are characteristic of saturated hydrocarbons. Addition reactions are characteristic of unsaturated hydrocarbons.
Important Carbon Compounds
Ethanol (Ethyl Alcohol) — C₂H₅OH
Preparation (Fermentation):
Sucrose (C₁₂H₂₂O₁₁) + H₂O →(Invertase) Glucose + Fructose (C₆H₁₂O₆)
C₆H₁₂O₆ →(Zymase) 2C₂H₅OH + 2CO₂ (Fermentation at 298–303 K, absence of air)
Physical Properties:
- Colourless liquid with a pleasant smell and burning taste
- Boiling point: 78.1°C; Freezing point: –118°C
- Density: 0.79 g/mL (lighter than water)
- Miscible with water in all proportions
- Neutral compound — no effect on litmus
Chemical Reactions:
Combustion:
C₂H₅OH + 3O₂ → 2CO₂ + 3H₂O + Heat (Blue flame)
Reaction with Sodium:
2C₂H₅OH + 2Na → 2C₂H₅ONa + H₂↑ (Hydrogen gas released with a pop sound)
Esterification (Reaction with Ethanoic Acid):
CH₃COOH + C₂H₅OH ⇌(Conc. H₂SO₄, Heat) CH₃COOC₂H₅ + H₂O (Ethyl ethanoate – sweet smell)
Dehydration (Reaction with Conc. H₂SO₄):
C₂H₅OH →(Conc. H₂SO₄, 443 K) CH₂=CH₂ + H₂O (Ethene)
Uses: Alcoholic beverages, solvent in paints/cosmetics/perfumes, antiseptic, power alcohol (80% ethanol + 20% petrol), antifreeze, manufacture of terylene and polythene, preservative for biological specimens.
Harmful Effects: Drunkenness; CNS depression (lack of coordination, mental confusion, drowsiness); long-term use damages the liver; absolute alcohol (pure ethanol) can be lethal even in small quantities.
Denatured Alcohol: Ethanol to which toxic/nauseating substances (methanol, pyridine) are added to prevent misuse. Industrial alcohol is coloured blue for identification. Methanol is extremely poisonous even small quantities can cause blindness or death by being oxidised to toxic methanal in the liver.
Ethanoic Acid (Acetic Acid) — CH₃COOH
Ethanoic acid is the second member of the carboxylic acid homologous series. A 5–8% solution of acetic acid in water is called vinegar, widely used as a food preservative.
Physical Properties:
- Colourless liquid with a strong, pungent smell and sour taste
- Boiling point: 391 K (118°C); Density: 1.08 g/mL at 273 K (heavier than water)
- Miscible with water (due to hydrogen bonding)
- Glacial acetic acid: Pure ethanoic acid that freezes at 289.6 K (16.6°C) into ice-like crystals
Chemical Reactions:
Esterification:
CH₃COOH + C₂H₅OH ⇌(Conc. H₂SO₄, Heat) CH₃COOC₂H₅ + H₂O
Saponification (Hydrolysis of Ester with Base):
CH₃COOC₂H₅ + NaOH → CH₃COONa + C₂H₅OH
Reaction with NaHCO₃:
NaHCO₃ + CH₃COOH → CH₃COONa + H₂O + CO₂↑
Reaction with Na₂CO₃:
Na₂CO₃ + 2CH₃COOH → 2CH₃COONa + H₂O + CO₂↑
Observation: Unlike strong acids (HCl), ethanoic acid is a weak acid. At equal molar concentrations, HCl has a lower pH (more acidic) than CH₃COOH.
Uses: Manufacture of dyes, perfumes, rayon; making vinegar; white lead for paints; coagulation of latex; preparation of cellulose acetate (photographic film); herbicide production; mordants in dyeing fabrics.
Soaps and Detergents
What is Soap?
A soap is the sodium or potassium salt of a long-chain fatty acid (carboxylic acid).
Examples: Sodium stearate (C₁₇H₃₅COONa), Sodium palmitate (C₁₅H₃₁COONa)
Soap solutions are basic in nature they turn red litmus paper blue.
Preparation of Soap (Saponification)
Soap is made by heating animal fats or vegetable oils (olive, castor, palm oil) with sodium hydroxide (NaOH) or potassium hydroxide (KOH).
Fat/Oil + NaOH → Soap + Glycerol
This process is called saponification.
Structure of a Soap Molecule
A soap molecule has two distinct ends:
- Hydrophobic "Tail": Long non-polar hydrocarbon chain — oil/grease loving, water hating
- Hydrophilic "Head": Ionic –COO⁻Na⁺ group — water loving, grease hating
Cleansing Action of Soap — Micelle Formation
When soap is added to water containing oily/greasy dirt:
- The hydrophobic tail of soap molecules dissolves in the oil/grease droplet.
- The hydrophilic head points outward into the water.
- Multiple soap molecules arrange around an oil droplet with tails pointing inward and heads pointing outward, forming a spherical cluster called a micelle.
- These micelles remain suspended in water (an emulsion) and are rinsed away during washing.
Disadvantages of Soap
Ineffective in Hard Water: Hard water contains Ca²⁺ and Mg²⁺ ions that react with soap to form an insoluble precipitate called scum (calcium stearate/magnesium stearate), wasting soap and leaving deposits on clothes and skin.
2C₁₇H₃₅COONa + Ca²⁺(aq) → (C₁₇H₃₅COO)₂Ca↓ + 2Na⁺
Ineffective in Acidic Medium: In acidic conditions, carboxylate ions (RCOO⁻) combine with H⁺ ions to form undissociated fatty acids, preventing micelle formation.
Synthetic Detergents
Synthetic detergents (called soapless soaps) are sodium salts of sulphonic acids (–SO₃H group), prepared from hydrocarbons derived from coal or petroleum.
Structure: They have the same hydrophobic tail and hydrophilic head as soap, but the head group is –SO₃⁻Na⁺ instead of COO⁻Na⁺.
Differences Between Soaps and Synthetic Detergents
| Feature | Soaps | Synthetic Detergents |
|---|---|---|
| Chemical nature | Sodium/potassium salts of long-chain fatty acids | Sodium salts of long-chain sulphonic acids |
| Ionic group | –COO⁻Na⁺ | –SO₃⁻Na⁺ |
| Raw material | Animal fats or vegetable oils | Hydrocarbons from coal/petroleum |
| Effectiveness in hard water | Reduced (forms scum) | Unaffected |
| Effectiveness in acidic medium | Reduced | Effective |
| Biodegradability | Fully biodegradable | Some are non-biodegradable |
| Dissolution in water | Slower | Faster |
| Examples | Sodium stearate, sodium palmitate | Sodium lauryl sulphate, sodium dodecyl benzene sulphonate |
Washing Powder Composition (approx. 15–30% detergent):
- Sodium sulphate + sodium silicate (keeps powder dry)
- Sodium tripolyphosphate/sodium carbonate (maintains alkalinity)
- Carboxy methyl cellulose (keeps dirt suspended)
- Sodium perborate (mild bleaching agent)
Carbon and Its Compounds Practice Questions with Detailed Solutions
What is catenation? Why does carbon show maximum catenation among all elements?
Answer:
Catenation is the property of an element to form chemical bonds with its own atoms, producing chains or rings of varying lengths and complexity.
Carbon shows the maximum catenation tendency in the periodic table for the following reasons:
Strong C–C Bond Energy: The carbon–carbon single bond (C–C) has a very high bond enthalpy (~347 kJ/mol), making carbon chains stable and resistant to breaking.
Tetravalency: Each carbon atom has 4 valence electrons available for bonding, allowing it to bond with up to 4 other carbon atoms simultaneously while still forming chains.
Small Atomic Size: Carbon's small size reduces steric repulsion between atoms in the chain, enabling stable, compact structures.
Silicon, which is in the same group as carbon, also shows catenation but to a much lesser extent because Si–Si bonds are weaker and more susceptible to oxidation.
Q2. Explain with a diagram the cleansing action of soap. What are micelles?
Answer:
Soap molecules have a dual nature:
Hydrophobic tail (long hydrocarbon chain, non-polar) — dissolves in oil/grease
Hydrophilic head (–COO⁻Na⁺, ionic) — dissolves in water
Mechanism of Cleansing:
When soap is dissolved in water and clothes are introduced, soap molecules surround oily dirt on the fabric surface.
The hydrophobic tails of soap molecules embed themselves into the grease/oil droplet, while the hydrophilic ionic heads remain in the aqueous phase.
As more soap molecules orient themselves this way around a single oil droplet, they form a spherical cluster called a micelle, with all hydrophobic tails pointing inward (toward oil) and all hydrophilic heads pointing outward (toward water).
These micelles form a stable emulsion in water.
Mechanical agitation (washing) breaks the micelles away from the fabric, and rinsing carries the emulsified dirt away with the water.
Micelle: A spherical aggregate of soap molecules in water, formed around an oil/grease droplet, with hydrophobic tails pointing inward and hydrophilic heads pointing outward.
Q3. Distinguish between saturated and unsaturated hydrocarbons, giving two examples of each.
Answer:
| Feature | Saturated Hydrocarbons | Unsaturated Hydrocarbons |
|---|---|---|
| Definition | Contain only single C–C bonds | Contain at least one C=C or C≡C bond |
| Also called | Alkanes | Alkenes (double bond) and Alkynes (triple bond) |
| General formula | CₙH₂ₙ₊₂ | CₙH₂ₙ (alkenes); CₙH₂ₙ₋₂ (alkynes) |
| Nature of flame | Clean blue flame (in excess O₂) | Luminous, smoky yellow flame |
| Typical reaction | Substitution reactions | Addition reactions |
| Examples | Methane (CH₄), Ethane (C₂H₆) | Ethene (C₂H₄), Ethyne (C₂H₂) |
Q4. What happens when ethanol reacts with sodium metal? Write the balanced chemical equation.
Answer:
When ethanol reacts with sodium metal, sodium ethoxide is formed and hydrogen gas is released.
2C₂H₅OH + 2Na → 2C₂H₅ONa + H₂↑
Observations:
Effervescence is observed as hydrogen gas is produced.
When a burning matchstick is brought near the gas, it burns with a distinctive "pop" sound — confirming the gas is hydrogen.
Significance: This reaction demonstrates that the –OH group in ethanol contains an acidic hydrogen (though much weaker than typical acids), which is replaced by the sodium metal. However, ethanol is a neutral compound and does not react with carbonates or hydrogen carbonates (unlike ethanoic acid).
Q5. What is esterification? Write the equation for the reaction between ethanol and ethanoic acid. What are the uses of esters?
Answer:
Esterification is the reaction between a carboxylic acid and an alcohol in the presence of concentrated sulphuric acid (H₂SO₄) as a catalyst/dehydrating agent to form an ester and water.
CH₃COOH + C₂H₅OH ⇌(Conc. H₂SO₄, Heat) CH₃COOC₂H₅ + H₂O
The product ethyl ethanoate (ethyl acetate) is a sweet-smelling ester.
The reverse reaction (breaking of ester in water) is called hydrolysis. When hydrolysis occurs in the presence of a base (like NaOH), it is called saponification the principle behind soap making.
Uses of Esters:
Naturally occurring esters give fruits their characteristic pleasant fragrances.
Used as solvents in adhesives, paints, and nail polish removers.
Used in the manufacture of perfumes, artificial flavours, and food additives.
Used in making plastics and fibres (e.g., polyester).
Q6. Why are soaps ineffective in hard water? How do synthetic detergents overcome this problem?
Answer:
Why soaps are ineffective in hard water:
Hard water contains dissolved calcium (Ca²⁺) and magnesium (Mg²⁺) ions. The carboxylate ions (RCOO⁻) of soap react with these ions to form insoluble calcium or magnesium salts:
2C₁₇H₃₅COONa + Ca²⁺(aq) → (C₁₇H₃₅COO)₂Ca↓ + 2Na⁺(aq)
This insoluble precipitate, called scum, sticks to clothes, skin, and utensils, reducing the soap's effectiveness. Large amounts of soap are wasted before it can perform any actual cleaning.
How synthetic detergents overcome this:
Synthetic detergents contain sulphonate groups (–SO₃⁻) instead of carboxylate groups (–COO⁻). The calcium and magnesium salts of sulphonic acids are soluble in water, so they do not form a scum in hard water. This means synthetic detergents retain their full cleansing ability regardless of water hardness. They can also be used in acidic solutions and sea water, where soaps fail.
Q7. What is a homologous series? State any four characteristics of homologous series.
Answer:
Homologous Series: A series of organic compounds having the same functional group, similar chemical properties, and the same general formula, in which successive members differ by a –CH₂ group (molecular mass difference of 14 u).
Four main point:
Common General Formula: All members can be described by one formula (e.g., CₙH₂ₙ₊₂ for alkanes).
Constant Difference of –CH₂: Successive members differ by one –CH₂ group, meaning the molecular mass increases by 14 u per step.
Similar Chemical Properties: All members react in the same way due to the same functional group (though reactivity may vary slightly with molecular size).
Gradual Change in Physical Properties: Melting point, boiling point, and density increase with increasing molecular mass in a predictable manner; physical state changes from gas (short chains) to liquid to solid (long chains).
Q8. Define isomerism. Draw the structures of the three isomers of pentane (C₅H₁₂).
Answer:
Isomerism is the phenomenon by which two or more compounds have the same molecular formula but different structural arrangements (structural formulae). Such compounds are called isomers.
Three isomers of Pentane (C₅H₁₂):
1. n-Pentane (Normal Pentane) - Straight chain:
CH3–CH2–CH2–CH2–CH3
2. Iso-Pentane (2-Methylbutane) - One branch:
CH3 | CH3–CH–CH2–CH3
3. Neo-Pentane (2,2-Dimethylpropane) - Two branches:
CH3 | CH3–C–CH3 | CH3
All three have the molecular formula C₅H₁₂ but differ in their structural arrangement, resulting in different physical properties (e.g., different boiling points).
Q9. What is the difference between a polar and non-polar covalent bond? Give one example of each.
Answer:
| Feature | Non-Polar Covalent Bond | Polar Covalent Bond |
|---|---|---|
| Formation | Between atoms of the same element or equal electronegativity | Between atoms of different electronegativities |
| Electron distribution | Shared pair lies exactly midway between both atoms | Shared pair is pulled closer to the more electronegative atom |
| Charge separation | No partial charges (δ⁺ or δ⁻) | Partial charges develop (δ⁺ on less electronegative atom, δ⁻ on more electronegative atom) |
| Dipole moment | Zero | Non-zero |
| Example | H–H (hydrogen molecule, H₂) | H–Cl (hydrogen chloride) |
In HCl: Chlorine is more electronegative than hydrogen. The shared pair is pulled toward Cl, making Cl partially negative (Cl^δ⁻) and H partially positive (H^δ⁺). This polarity makes HCl a polar molecule with a non-zero dipole moment.
Q10. Explain the oxidation of ethanol to ethanoic acid. Which reagents are used? Write the chemical equation.
Answer:
Ethanol (C₂H₅OH) can be oxidised to ethanoic acid (CH₃COOH) using strong oxidising agents:
Reagents:
- Alkaline potassium permanganate (KMnO₄) + Heat
- Acidified potassium dichromate (K₂Cr₂O₇/H₂SO₄) + Heat
Equation:
CH₃CH₂OH + 2[O] →(Alkaline KMnO₄, Heat) CH₃COOH + H₂O
The [O] represents nascent oxygen from the oxidising agent.
Laboratory Evidence (Activity):
- When alkaline KMnO₄ is added drop by drop to ethanol, the purple colour of KMnO₄ disappears initially (as ethanol is oxidised and KMnO₄ is consumed/reduced).
- Once all the ethanol is converted to ethanoic acid, additional KMnO₄ retains its purple colour, confirming the reaction is complete.
Biological Context: In the human body, ethanol is oxidised to ethanal (acetaldehyde) by the enzyme alcohol dehydrogenase, then to ethanoic acid by aldehyde dehydrogenase. This metabolic pathway in the liver explains the toxic effects of alcohol, particularly from methanol, which produces toxic methanal.
Carbon and Its Compounds Chapter Summary
- Carbon's unique properties — tetravalency, catenation, isomerism, and tendency to form multiple bonds — account for the existence of over 3 million organic compounds.
- Covalent bonds form by the sharing of electrons; carbon forms only covalent bonds, never ionic bonds under normal conditions.
- The three allotropes of carbon — diamond (hardest, non-conductor), graphite (soft, conductor), and fullerene (cage-like, semiconductor) — illustrate how structural arrangement determines physical properties.
- Hydrocarbons are classified as saturated (alkanes, single bonds) and unsaturated (alkenes with double bonds; alkynes with triple bonds). Homologous series group compounds by shared structure and formula.
- Combustion (oxidation in oxygen) is the key chemical property, producing CO₂ and H₂O. Saturated hydrocarbons burn cleanly; unsaturated ones produce soot.
- Ethanol and ethanoic acid are the two most important carbon compounds in daily life, involved in food, medicine, and industry.
- Soaps (saponification products, basic) work via micelle formation but fail in hard or acidic water. Synthetic detergents overcome this limitation but raise environmental concerns.
- Esterification (acid + alcohol → ester + water) and saponification (ester + base → acid salt + alcohol) are reversible reactions of fundamental industrial importance.
