Acids, Bases and Salts Class 10 Notes: The chapter acids bases and salts class 10 is one of the most important topics in Science. It helps students understand the basic chemical nature of substances that we use in daily life, such as lemon juice, soap, baking soda, and common salt.
Acids are substances that produce hydrogen ions (H⁺) in aqueous solution, while bases produce hydroxide ions (OH⁻). When an acid reacts with a base, it forms salt and water. This reaction is called neutralization reaction.
These acids, bases and salts class 10 notes explain important concepts like pH scale, chemical properties of acids and bases, indicators such as litmus, phenolphthalein and methyl orange, and the strength of acids and alkalis.
Students also learn about strong and weak acids, corrosive nature of acids, and uses of common salts like sodium chloride and baking soda. Sometimes students think all acids are dangerous, but that is not fully true because many acids are found in food items also.
To prepare well for exams, learners should practice acid bases and salts class 10 questions and answers and revise from a reliable class 10 acids, bases and salts notes pdf.
These acids bases and salts class 10 notes are designed in simple language so that every student can easily understand the concepts and build a strong foundation in chemistry.
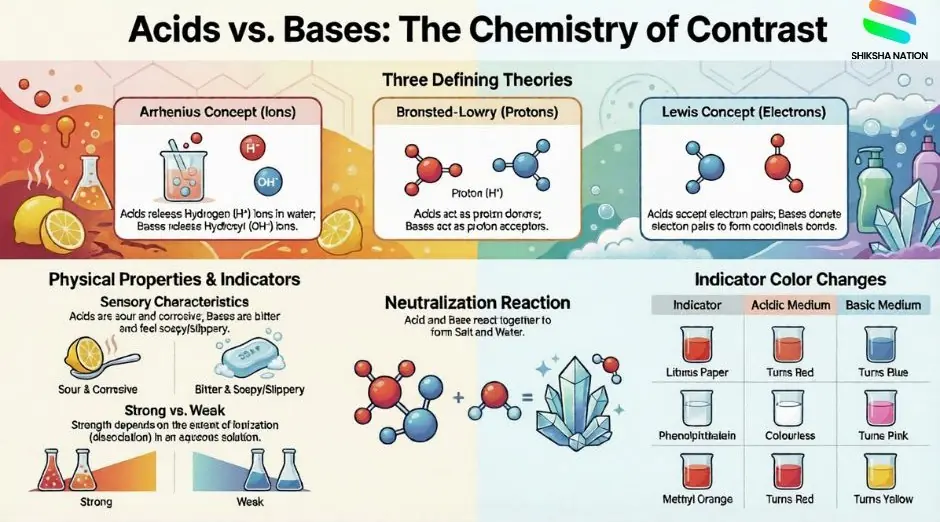
What is Acids, Bases and Salts?
Acids and bases are among the most fundamental concepts in chemistry. Historically, acids were identified by their sour taste and their ability to dissolve certain metals, affect vegetable dyes, and react with metals to produce gases. Bases were recognised by their bitter taste and soapy touch.
The scientific study of acids and bases began with Svante Arrhenius between 1880–1890, who proposed the first modern theory of acids and bases based on ionisation in water. This was followed by the Bronsted-Lowry concept in 1923 and the even broader Lewis concept, which remains the most comprehensive acid-base theory to date.
When an acid reacts with a base, a neutralisation reaction occurs:
Acid + Base → Salt + Water HCl (aq) + NaOH (aq) → NaCl (aq) + H₂O (l)
The strength of any acidic or basic solution is conveniently expressed using a scale called the pH scale, which is based on the concentration of H⁺ (or H₃O⁺) ions in solution.
CBSE Class 10 Science Acids Bases and Salts Notes PDF Download
Fill the form to download this PDF
Theories of Acids and Bases
Arrhenius Theory (1887)
The Swedish chemist Svante Arrhenius proposed the first modern definition of acids and bases based on their behaviour in aqueous solutions.
Arrhenius Acid:
A substance that releases one or more free hydrogen ions (H⁺) when dissolved in water.
Examples:
- HCl (aq) → H⁺ (aq) + Cl⁻ (aq) (Strong acid – complete dissociation)
- HNO₃ (aq) → H⁺ (aq) + NO₃⁻ (aq) (Strong acid)
- CH₃COOH (aq) ⇌ CH₃COO⁻ (aq) + H⁺ (aq) (Weak acid – partial dissociation)
Arrhenius Base:
A hydroxyl-containing compound that releases free hydroxide ions (OH⁻) when dissolved in water.
Examples:
- NaOH (aq) → Na⁺ (aq) + OH⁻ (aq) (Strong base)
- KOH (aq) → K⁺ (aq) + OH⁻ (aq) (Strong base)
- NH₄OH ⇌ NH₄⁺ (aq) + OH⁻ (aq) (Weak base)
Neutralisation (Arrhenius):
H⁺ (aq) + OH⁻ (aq) → H₂O (l)
Limitation: The Arrhenius theory applies only to aqueous solutions and cannot explain acid-base behaviour in non-aqueous solvents.
Bronsted-Lowry Theory (1923)
In 1923, J.N. Bronsted and J.M. Lowry independently proposed a more general definition:
Bronsted-Lowry Acid = A proton (H⁺) donor
Bronsted-Lowry Base = A proton (H⁺) acceptor
This theory introduces the important concept of conjugate acid-base pairs.
Example – Ionisation of HCl in water:
HCl (aq) + H₂O (l) → H₃O⁺ (aq) + Cl⁻ (aq)
(Acid) (Base) (Conjugate Acid) (Conjugate Base)
Here:
- HCl donates a proton → it is the acid
- H₂O accepts the proton → it is the base
- H₃O⁺ (hydronium ion) is the conjugate acid of water
- Cl⁻ is the conjugate base of HCl
More Examples:
| Reaction | Acid | Base | Conjugate Acid | Conjugate Base |
|---|---|---|---|---|
| HNO₃ + H₂O ⇌ H₃O⁺ + NO₃⁻ | HNO₃ | H₂O | H₃O⁺ | NO₃⁻ |
| HCN + H₂O ⇌ H₃O⁺ + CN⁻ | HCN | H₂O | H₃O⁺ | CN⁻ |
| H₂SO₄ + H₂O ⇌ H₃O⁺ + HSO₄⁻ | H₂SO₄ | H₂O | H₃O⁺ | HSO₄⁻ |
| H₂O + NH₃ ⇌ NH₄⁺ + OH⁻ | H₂O | NH₃ | NH₄⁺ | OH⁻ |
Amphoteric/Amphiprotic Species: Species that can act as both acid and base depending on conditions are called amphoteric or amphiprotic. Examples: H₂O and NH₃.
Advantage over Arrhenius: This theory extends to non-aqueous solvents and explains ionic species acting as acids or bases.
Lewis Theory (Most Advanced)
Proposed by G.N. Lewis, this is the most comprehensive and generalised concept of acids and bases. It does not require hydrogen as an essential component.
Lewis Acid = Electron pair acceptor (has empty orbitals) Lewis Base = Electron pair donor (has lone pair of electrons)
General Reaction:
A⁺ (empty orbital) + :B (lone pair) → B: → A (coordinate bond formed)
Examples:
| Lewis Acid | Lewis Base | Product |
|---|---|---|
| H⁺ | :NH₃ | NH₄⁺ |
| BF₃ | :NH₃ | H₃N→BF₃ |
| AlCl₃ | :Cl⁻ | AlCl₄⁻ |
Types of Lewis Acids:
- Molecules with incomplete octet at central atom: BF₃, AlCl₃, FeCl₃
- Simple cations (electron deficient): Na⁺, K⁺, H⁺, Ag⁺
- Molecules that can expand their octet: SiF₄, SnCl₄
Types of Lewis Bases:
- Neutral molecules with at least one lone pair: NH₃, H₂O
- Anions with negative charge: F⁻, Cl⁻, Br⁻, OH⁻
- Molecules with π bonds: H₂C=CH₂, HC≡CH
Insight: Sn⁴⁺ is a stronger Lewis acid than Sn²⁺ because it has a greater number of vacant orbitals.
Basic Properties of Acids and Bases
Properties of Acids
| Property | Description |
|---|---|
| Taste | Sour (e.g., lemon juice, vinegar) |
| Litmus | Turns blue litmus red |
| Phenolphthalein | Remains colourless |
| Methyl orange | Turns red |
| Electrical conductivity | Conduct electricity (produce ions) |
| Reaction with metals | Produce hydrogen gas |
| Reaction with bases | Form salt and water (neutralisation) |
Properties of Bases
| Property | Description |
|---|---|
| Taste | Bitter |
| Touch | Soapy/slippery |
| Litmus | Turns red litmus blue |
| Phenolphthalein | Turns pink |
| Methyl orange | Turns yellow |
| Electrical conductivity | Conduct electricity (produce OH⁻ ions) |
Remember: The term "acid" is derived from the Latin word "acidus" meaning sour to taste.
Classification of Acids
A. Based on Source
| Type | Description | Examples |
|---|---|---|
| Mineral/Inorganic Acids | Obtained from minerals/rocks | HCl, H₂SO₄, HNO₃, H₂CO₃, H₃PO₄ |
| Organic Acids | Obtained from plants/animals | HCOOH (Formic), CH₃COOH (Acetic), C₆H₅COOH (Benzoic), C₆H₈O₇ (Citric) |
B. Based on Basicity (Number of H⁺ ions released)
| Type | H⁺ ions produced | Examples |
|---|---|---|
| Monobasic | 1 | HCl, HBr |
| Dibasic | 2 | H₂SO₄, H₂CO₃ |
| Tribasic | 3 | H₃PO₄, Citric acid |
C. Based on Strength (Degree of Ionisation)
| Type | Ionisation | Examples |
|---|---|---|
| Strong Acids | Complete | HCl, H₂SO₄, HNO₃ |
| Weak Acids | Partial/Incomplete | CH₃COOH, HCOOH, H₂CO₃, H₃PO₄ |
Key Fact: In general, mineral acids are strong acids while organic acids are weak acids.
D. Based on Concentration
- Concentrated acid – less water, more acid
- Dilute acid – more water, less acid
E. Based on Oxygen Content
- Binary acids (Hydroacids) – H combined with one other element: HCl, HCN
- Oxy acids – contain oxygen: HNO₃, H₂SO₄
Classification of Bases
A. Based on Acidity (OH⁻ ions released)
| Type | OH⁻ ions | Examples |
|---|---|---|
| Monoacidic | 1 | NaOH, KOH |
| Diacidic | 2 | Ca(OH)₂, Mg(OH)₂ |
| Triacidic | 3 | Al(OH)₃, Fe(OH)₃ |
B. Based on Strength
| Type | Ionisation | Examples |
|---|---|---|
| Strong Bases | Complete | NaOH, KOH, Ba(OH)₂ |
| Weak Bases | Incomplete | Mg(OH)₂, NH₄OH, Cu(OH)₂ |
Important Rule: All alkalis are bases but all bases are not alkalis. Alkalis are bases that are soluble in water (e.g., NaOH, KOH, Ca(OH)₂).
Note: Both NaOH and KOH are deliquescent they absorb moisture from the air and get liquefied.
Chemical Properties - Reactions of Acids and Bases
Indicators
Indicators are substances that show different colours in acidic and basic media. They are coloured organic substances, either naturally extracted or synthetically prepared.
Common Acid-Base Indicators
| Indicator | Neutral | Acidic Medium | Basic Medium |
|---|---|---|---|
| Litmus | Purple | Red | Blue |
| Phenolphthalein | Colourless | Colourless | Pink |
| Methyl orange | Orange | Red | Yellow |
| Red cabbage juice | Purple | Red/Pink | Green |
| Turmeric | Yellow | Yellow | Reddish/Brown |
- Natural indicators: Litmus (from lichen/Thallophyta), turmeric, red cabbage
- Synthetic indicators: Phenolphthalein, methyl orange
- Olfactory indicators: Onion, vanilla essence, clove oil – change odour in acid/base
Reaction of Acids with Metals
Active metals displace hydrogen from dilute acids:
Metal + Dilute Acid → Metal Salt + Hydrogen↑
2Na + 2HCl → 2NaCl + H₂↑
Mg + H₂SO₄ → MgSO₄ + H₂↑
Zn + H₂SO₄ → ZnSO₄ + H₂↑
Fe + H₂SO₄ → FeSO₄ + H₂↑
Active metals: Na, K, Ca, Mg, Zn, Fe
Test for H₂ gas: Burns with a characteristic "pop" sound when a burning candle is brought near it.
Note: Copper, mercury, and silver do not react with dilute HCl or H₂SO₄.
Reaction of Metals with Bases
Metals like zinc, tin, and aluminium react with strong alkalis to release hydrogen:
Zn + 2NaOH → Na₂ZnO₂ + H₂↑ (sodium zincate)
Sn + 2NaOH → Na₂SnO₂ + H₂↑ (sodium stannite)
2Al + 2NaOH + 2H₂O → 2NaAlO₂ + 3H₂↑ (sodium meta aluminate)
Action of Acids with Metal Oxides
Metal oxides (basic in nature) react with acids to form salt and water:
Metal Oxide + Acid → Salt + Water
ZnO + 2HCl → ZnCl₂ + H₂O
MgO + H₂SO₄ → MgSO₄ + H₂O
CuO + 2HCl → CuCl₂ + H₂O (black → bluish green)
CaO + 2HCl → CaCl₂ + H₂O
Action of Bases with Non-Metallic Oxides
Bases react with non-metallic oxides (e.g., CO₂, SO₂, SO₃) to form salt and water:
2NaOH + CO₂ → Na₂CO₃ + H₂O
CO₂ + Ca(OH)₂ → CaCO₃↓ + H₂O (lime water turns milky)
2KOH + CO₂ → K₂CO₃ + H₂O
Note: CO₂, SO₂, SO₃ are non-metallic oxides and are acidic in nature.
Reaction of Acids with Metal Carbonates and Bicarbonates
Metal Carbonate/Bicarbonate + Acid → Salt + CO₂↑ + Water
CaCO₃ + 2HCl → CaCl₂ + H₂O + CO₂↑
Na₂CO₃ + H₂SO₄ → Na₂SO₄ + H₂O + CO₂↑
2NaHCO₃ + H₂SO₄ → Na₂SO₄ + 2H₂O + 2CO₂↑
MgCO₃ + 2HNO₃ → Mg(NO₃)₂ + H₂O + CO₂↑
MgCO₃ + 2HCl → MgCl₂ + H₂O + CO₂↑
Lime Water Test for CO₂: CO₂ turns lime water milky due to formation of CaCO₃. Excess CO₂ clears the milkiness again by forming soluble Ca(HCO₃)₂.
Interesting fact: Egg shells, limestone, chalk, and marble are all different forms of calcium carbonate (CaCO₃).
Neutralisation Reaction
When an acid and a base react in aqueous solution, they form a salt and water. This is called a neutralisation reaction.
Acid + Base → Salt + Water
HCl + NaOH → NaCl + H₂O
HNO₃ + KOH → KNO₃ + H₂O
HCl + NH₄OH → NH₄Cl + H₂O
At the ionic level:
H⁺ (aq) + OH⁻ (aq) → H₂O (l)
Practical Applications of Neutralisation:
- Antacid tablets (NaHCO₃, Mg(OH)₂) neutralise excess HCl in the stomach
- Bee/ant stings (formic acid) are neutralised by rubbing with soap (NaOH)
- Wasp stings (alkaline) are neutralised by vinegar (acetic acid)
- Soil acidity from acid rain is neutralised by slaked lime (calcium hydroxide)

Dissociation of Acids and Bases
Dissociation of Acids
Hydrogen ions (H⁺) don't exist independently in solution. They combine with water to form hydronium ions (H₃O⁺):
H⁺ + H₂O → H₃O⁺
HCl + H₂O → H₃O⁺ + Cl⁻
H₂SO₄ + 2H₂O → 2H₃O⁺ + SO₄²⁻
Important: Not all hydrogen-containing compounds are acids. Ethyl alcohol (C₂H₅OH) and glucose (C₆H₁₂O₆) do not produce H⁺ ions and hence are not acids.
Basicity of an Acid = Number of replaceable H⁺ ions per molecule
| Acid | Reaction | Basicity |
|---|---|---|
| HCl | HCl ⇌ H⁺ + Cl⁻ | 1 |
| H₂SO₄ | H₂SO₄ ⇌ 2H⁺ + SO₄²⁻ | 2 |
| H₃PO₄ | H₃PO₄ ⇌ 3H⁺ + PO₄³⁻ | 3 |
Dissociation of Bases
NaOH (s) → Na⁺ (aq) + OH⁻ (aq)
Ca(OH)₂ (s) → Ca²⁺ (aq) + 2OH⁻ (aq)
Acidity of a Base = Number of replaceable OH⁻ groups per molecule
| Base | Reaction | Acidity |
|---|---|---|
| NaOH | NaOH ⇌ Na⁺ + OH⁻ | 1 |
| Ca(OH)₂ | Ca(OH)₂ ⇌ Ca²⁺ + 2OH⁻ | 2 |
| Fe(OH)₃ | Fe(OH)₃ ⇌ Fe³⁺ + 3OH⁻ | 3 |
Common Property of Acids and Bases: Both produce ions in aqueous solution and thus conduct electricity. Ethanol and glucose, which do not produce ions, are non-conductors.
Dilution of Acids and Bases
- Dilution is exothermic heat is released when acid/base is dissolved in water.
- Safety Rule: Always add acid to water, never water to acid. Adding water to concentrated acid causes violent reaction, splashing, and may crack glassware.
The pH Scale
The pH scale was introduced by Danish biochemist S. Sorensen. The term 'p' comes from the German word "potenz" (meaning power).
Mathematical Definition:
pH = –log[H⁺]
pH Scale Summary
| pH Range | Nature of Solution | Example |
|---|---|---|
| pH = 0–6 | Acidic (decreasing pH = stronger acid) | HCl, H₂SO₄ |
| pH = 7 | Neutral | Pure water |
| pH = 8–14 | Basic (increasing pH = stronger base) | NaOH, KOH |
Relation Between pH and Ion Concentrations
- Neutral: pH = 7, [H⁺] = 10⁻⁷ M
- Acidic: pH < 7, [H⁺] > 10⁻⁷ M
- Basic: pH > 7, [H⁺] < 10⁻⁷ M
pOH:
pOH = –log[OH⁻] pH + pOH = 14
Ionic Product of Water (Kw)
Pure water self-ionises to a tiny extent:
H₂O + H₂O ⇌ H₃O⁺ + OH⁻
Kw = [H₃O⁺][OH⁻] = 1.0 × 10⁻¹⁴ (at 25°C / 298 K)
Solved Examples
Q1. pH of 0.0001 M HCl:
[H⁺] = 10⁻⁴ M → pH = 4
Q2. pH of 0.001 M NaOH:
[OH⁻] = 10⁻³ M → pOH = 3 → pH = 14 – 3 = 11
Q3. pH of 0.1 M HCl:
[H⁺] = 0.1 = 10⁻¹ → pH = 1
Importance of pH in Everyday Life
i. pH of the Soil
Different plants require different pH ranges. Acidic soil (from rotting vegetation or iron minerals) can be corrected by "liming" (adding Ca(OH)₂, CaO, or CaCO₃). Alkaline soil can be treated with compost/manure.
| Plant | Preferred pH |
|---|---|
| Potato | 4.5–6.0 |
| Carrot, Sweet Potato | 5.5–6.5 |
| Cauliflower, Garlic, Tomato | 5.5–7.5 |
| Onion, Cabbage | 6.0–7.5 |
ii. pH in Our Digestive System
The stomach produces HCl for digestion. Indigestion (excess acid) causes pain. Antacids (Mg(OH)₂ – milk of magnesia, NaHCO₃) neutralise excess acid and provide relief.
iii. Tooth Decay and pH
Tooth enamel (calcium phosphate) begins to corrode when mouth pH falls below 5.5. Bacteria decompose food sugars to produce acids. Basic toothpastes neutralise the acid and prevent decay. Saliva (alkaline) also partially neutralises mouth acids.
iv. pH of Blood
Human blood has a pH between 7.36 and 7.42, maintained by buffer systems (hydrogen carbonates + carbonic acid) in the blood.
v. Acid Rain
When rainwater pH drops below 5.6, it is called acid rain, which harms aquatic life and soil.
vi. Self-Defence in Nature
- Bee stings → inject acid → treated with mild base (baking soda)
- Wasp stings → inject alkali → treated with vinegar (acetic acid)
- Stinging nettles → inject methanoic acid → treated by rubbing with dock plant leaves
More About Salts
Salts are ionic compounds formed by the reaction of an acid with a base, consisting of:
- Cation – positive ion (from the base)
- Anion – negative ion (from the acid)
pH of Salts
| Salt Type | Example | pH |
|---|---|---|
| Strong acid + Strong base | NaCl | = 7 (Neutral) |
| Strong acid + Weak base | NH₄Cl | < 7 (Acidic) |
| Weak acid + Strong base | Na₂CO₃ | > 7 (Basic) |
| Weak acid + Weak base | CH₃COONH₄ | ≈ 7 |
Family of Salts
Salts are grouped into families based on their common ion:
- Sulphate family: K₂SO₄, Na₂SO₄, CaSO₄, MgSO₄, CuSO₄
- Chloride family: NaCl, NH₄Cl
- Sodium family: Na₂SO₄, NaCl, NaNO₃, Na₂CO₃
Formation of Salts
| Method | Reaction |
|---|---|
| Acid + Base | NaOH + HCl → NaCl + H₂O |
| Metal + Acid | Zn + H₂SO₄ → ZnSO₄ + H₂↑ |
| Metal carbonate + Acid | CaCO₃ + 2HCl → CaCl₂ + H₂O + CO₂↑ |
| Metal + Alkali | 2NaOH + Zn → Na₂ZnO₂ + H₂↑ |
Important Salts Detailed Study
Sodium Chloride (Common Salt – NaCl)
- Formed from HCl + NaOH → neutral salt (pH = 7)
- Found in sea water; extracted as rock salt by evaporation
- Raw material for NaOH, NaHCO₃, Na₂CO₃, bleaching powder
Sodium Hydroxide – NaOH (Caustic Soda)
Produced by Chloralkali process (electrolysis of brine):
2NaCl (aq) + 2H₂O (l) → 2NaOH (aq) + Cl₂↑ + H₂↑
- At cathode: H₂ gas
- At anode: Cl₂ gas
- Near cathode: NaOH solution forms
Uses: Soaps, detergents, paper making, artificial fibres, de-greasing metals.
Bleaching Powder – CaOCl₂ (Calcium Oxychloride)
Preparation: Chlorine gas passed over dry slaked lime:
Ca(OH)₂ + Cl₂ → CaOCl₂ + H₂O
Properties:
- Yellowish-white powder with strong chlorine smell
- Soluble in water (with residue of lime)
- Deteriorates on exposure to air by reacting with CO₂: CaOCl₂ + CO₂ → CaCO₃ + Cl₂↑
- With dilute acid, releases "available chlorine": CaOCl₂ + H₂SO₄ → CaSO₄ + H₂O + Cl₂↑
Disinfection mechanism: CaOCl₂ + water → hypochlorous acid (HOCl) → germicidal action
Uses:
- Bleaching cotton, linen, wood pulp, paper
- Sterilisation/disinfection of drinking water
- Making wool unshrinkable
- Oxidising agent in chemical manufacturing
Baking Soda – NaHCO₃ (Sodium Hydrogen Carbonate)
Production:
NaCl + H₂O + CO₂ + NH₃ → NH₄Cl + NaHCO₃
Properties:
- White crystalline solid, sparingly soluble in water
- Aqueous solution is weakly alkaline (due to hydrolysis)
- Thermal decomposition: 2NaHCO₃ → Na₂CO₃ + H₂O + CO₂↑ (on heating)
- Reaction with acids: NaHCO₃ + HCl → NaCl + H₂O + CO₂↑2NaHCO₃ + H₂SO₄ → Na₂SO₄ + 2H₂O + 2CO₂↑
Uses:
- Baking powder = NaHCO₃ + tartaric acid → CO₂ released makes bread/cake soft and spongy
- Antacid – neutralises excess HCl in stomach: NaHCO₃ + HCl → NaCl + H₂O + CO₂
- Soda-acid fire extinguishers – NaHCO₃ + H₂SO₄ → CO₂ pushes water out to extinguish fire
- Mild antiseptic for infections
Washing Soda – Na₂CO₃·10H₂O (Sodium Carbonate)
Manufactured by the Solvay (Ammonia-soda) process:
Step 1: NaCl + H₂O + CO₂ + NH₃ → NaHCO₃ + NH₄Cl
Step 2: 2NaHCO₃ → Na₂CO₃ + H₂O + CO₂↑ (heating)
Step 3: Na₂CO₃ + 10H₂O → Na₂CO₃·10H₂O (crystallisation)
Anhydrous sodium carbonate = Soda ash (Na₂CO₃)
Uses:
- Washing and cleaning clothes
- Softening hard water
- Manufacturing detergents, glass, paper, paint
- Textile industry – helps dyes bond to fabric
Difference between Baking Soda and Washing Soda:
- Baking soda: NaHCO₃ (mild, used in food/antacids)
- Washing soda: Na₂CO₃·10H₂O (stronger base, used in cleaning)
- They cannot substitute each other; washing soda in baking causes bitterness.
Plaster of Paris – CaSO₄·½H₂O
Plaster of Paris is the hemihydrate of calcium sulphate.
Preparation: Heating gypsum (CaSO₄·2H₂O) at 373 K (100°C):
2CaSO₄·2H₂O → (CaSO₄)₂·H₂O + 3H₂O
OR
CaSO₄·2H₂O → CaSO₄·½H₂O + 3/2 H₂O
Caution: If heated above 373 K, anhydrous CaSO₄ (Dead burnt plaster) forms, which does NOT set with water.
Setting of Plaster of Paris:
CaSO₄·½H₂O + 3/2 H₂O → CaSO₄·2H₂O (Gypsum – hard mass)
During setting, a slight volume expansion (~1%) occurs, ensuring the material fills the mould completely and gives a sharp impression.
Properties:
- White, odourless powder
- Absorbs water and releases heat at room temperature
- With 50% water by mass, forms a plastic mass that sets hard within minutes
Uses:
- Medical use – setting fractured/broken bones; immobilising injured parts
- Pottery and ceramics – producing moulds
- Making statues, models, decorative items
- Fire-proofing material and making chalks
- Sealing air gaps in laboratory apparatus
- Filling small gaps in walls and roofs
Water of Crystallisation?
Water of crystallisation is the definite number of water molecules loosely combined with one formula unit of an ionic solid in its crystalline form. These water molecules contribute to the crystal's shape and colour.
| Hydrated Salt | Common Name | Water Molecules |
|---|---|---|
| CuSO₄·5H₂O | Blue Vitriol / Copper Sulphate | 5 |
| Na₂CO₃·10H₂O | Washing Soda | 10 |
| Na₂SO₄·10H₂O | Glauber's Salt | 10 |
| MgSO₄·7H₂O | Epsom Salt | 7 |
| CaSO₄·2H₂O | Gypsum | 2 |
| CaSO₄·½H₂O | Plaster of Paris | ½ |
On heating to 100°C, copper sulphate (blue crystals, CuSO₄·5H₂O) loses water to become anhydrous copper sulphate (white powder, CuSO₄).
Related Terms
| Term | Definition | Examples |
|---|---|---|
| Efflorescence | Spontaneous loss of water of crystallisation when exposed to air | Na₂CO₃·10H₂O, CuSO₄·5H₂O |
| Deliquescence | Absorption of moisture from air to form a solution | NaOH, KOH, CaCl₂ |
| Hygroscopic | Absorption of moisture without change of physical state | Anhydrous Na₂CO₃, Conc. H₂SO₄ |
Class 10 Science Acid Bases and Salts Quick Revision Points
- Acids release H⁺ ions in aqueous solution; bases release OH⁻ ions
- Arrhenius theory is limited to aqueous solutions
- Bronsted-Lowry theory defines acids as proton donors and bases as proton acceptors
- Lewis theory defines acids as electron pair acceptors and bases as electron pair donors
- Litmus is a natural indicator from lichen (Thallophyta)
- Phenolphthalein and methyl orange are synthetic (laboratory) indicators
- Onion, vanilla, clove oil are olfactory indicators
- Active metals react with dilute acids to release H₂ gas
- Cu, Hg, Ag do not react with dilute HCl or H₂SO₄
- CO₂, SO₂, SO₃ are non-metallic oxides – acidic in nature
- Metal oxides are basic in nature
- All bases produce OH⁻ ions; all acids produce H⁺ ions in solution
- Aqueous solutions of acids and bases conduct electricity (due to ions)
- Ethanol and glucose do not conduct electricity
- pH = 7 (neutral), < 7 (acidic), > 7 (basic)
- pH + pOH = 14; Kw = 1.0 × 10⁻¹⁴ at 25°C
- Human body functions optimally at pH 7.0–7.8
- Tooth decay begins when mouth pH falls below 5.5
- Acid rain: pH below 5.6
- Blood pH: 7.36–7.42 (maintained by buffers)
- All alkalis are bases but not all bases are alkalis
- NaHCO₃ (baking soda) releases CO₂ on heating or reacting with acid
- CaOCl₂ (bleaching powder) releases available chlorine responsible for bleaching
- Washing soda = Na₂CO₃·10H₂O; used for water softening
- Plaster of Paris sets by reabsorbing water to form gypsum
- Dead burnt plaster (anhydrous CaSO₄) does not set with water
