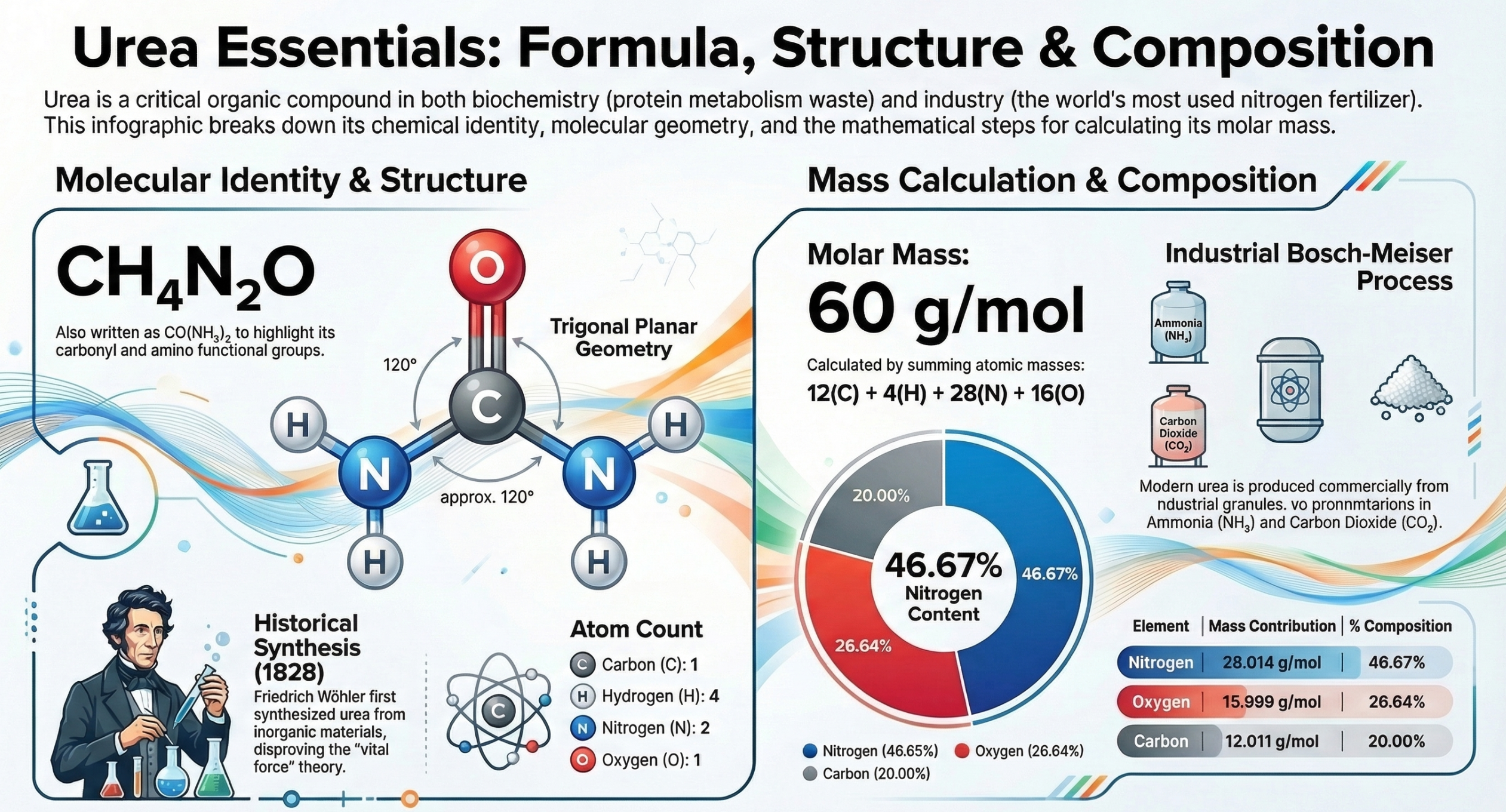
What Is Urea?
Urea is an organic compound that serves as the primary nitrogen-containing waste product of protein metabolism in mammals. It is excreted in urine, making it a key compound in biochemistry and renal physiology.
In industry, urea is the world’s most widely used nitrogen-based fertilizer, supplying 46% nitrogen by weight. It is also used in plastics, resins, animal feed supplements, and medical diagnostics.

Identity Facts About Urea:
- Also known as: Carbamide
- Discovered by: Friedrich Wohler (1828) first organic compound synthesized from inorganic materials
- Physical form: White crystalline solid at room temperature
- Taste: Slightly salty
- Solubility: Highly soluble in water
- State at Room Temperature: Solid
Urea Formula
The Chemical Formula of Urea:
CH4N2O (also written as CO(NH2)2 or H2NCONH2 or (NH2)2CO)
The formula CH4N2O tells us the exact number of each type of atom present in one molecule of urea:
| Element |
Symbol |
Number of Atoms |
Atomic Mass (g/mol) |
Total Mass Contribution |
| Carbon |
C |
1 |
12.011 |
12.011 |
| Hydrogen |
H |
4 |
1.008 |
4.032 |
| Nitrogen |
N |
2 |
14.007 |
28.014 |
| Oxygen |
O |
1 |
15.999 |
15.999 |
| TOTAL |
— |
8 atoms |
— |
60.056 g/mol |
The formula CO(NH2)2 is the expanded or functional form that shows the two amino groups (NH2) attached to a carbonyl group (C=O). Both notations represent the same compound.
Urea Formula Name (IUPAC & Common Names)
| Name Type |
Name |
Context of Use |
| IUPAC Name |
Carbonamide (or Diaminomethanone) |
Formal chemistry, research papers |
| Common Name |
Urea |
Everyday chemistry, exams, textbooks |
| Alternative Common Name |
Carbamide |
Pharmaceutical, biochemistry contexts |
| Systematic Name |
Diaminoformaldehyde |
Older literature |
| Biochemistry Name |
Urea (end product of urea cycle) |
Biology, physiology, nephrology |
The IUPAC name carbonamide and the common name urea are both accepted. Most Indian board exams and competitive exams use the name urea with the formula CH4N2O or CO(NH2)2.
Urea Molecular Formula
What the Molecular Formula
The molecular formula CH4N2O reveals:
- C = 1 carbon atom in the molecule
- H = 4 hydrogen atoms (2 from each NH2 group)
- N = 2 nitrogen atoms (one in each amino group)
- O = 1 oxygen atom (in the carbonyl group C=O)
Empirical Formula vs. Molecular Formula of Urea
| Formula Type |
Formula |
Meaning |
| Molecular Formula |
CH4N2O |
Exact number of each atom per molecule |
| Empirical Formula |
CH4N2O |
Simplest whole-number ratio (same as molecular for urea) |
| Structural Formula |
H2N-CO-NH2 |
Shows how atoms are bonded |
| Condensed Formula |
CO(NH2)2 |
Shows functional groups clearly |
| Lewis Structure Formula |
(NH2)2C=O |
Shows bonding and lone pairs |
Important Note:
For urea, the empirical formula and molecular formula are the SAME — CH4N2O — because the ratio of atoms C:H:N:O = 1:4:2:1 is already in its simplest form and cannot be reduced further.
Urea Structural Formula & Lewis Structure
Structural Formula of Urea
Structural Formula of Urea
O = H2N — C — NH2
(carbonyl carbon bonded to two amino groups)
Explanation of Urea’s Structure
- Central Carbon Atom: sp2 hybridized, forms 3 sigma bonds
- Carbonyl Group (C=O): A double bond between carbon and oxygen this is the defining feature
- Two Amino Groups (NH2): Each nitrogen has 2 hydrogen atoms and one lone pair
- Bond Angles: Approximately 120 degrees around the central carbon (trigonal planar geometry)
- Resonance: Urea exhibits resonance; the C-N bonds have partial double bond character
Geometry & Hybridization
| Feature |
Detail |
| Geometry |
Trigonal planar around central carbon |
| Hybridization of C |
sp2 |
| Hybridization of N |
sp3 (but with partial sp2 due to resonance) |
| Bond Angle (N-C-N) |
~116 degrees |
| Bond Angle (N-C-O) |
~122 degrees |
| C=O Bond Length |
1.26 A (shorter than typical C-O, longer than pure C=O due to resonance) |
| C-N Bond Length |
1.34 A (shorter than typical C-N, showing partial double bond character) |
Step-by-Step Calculation of Urea Formula Molecular Mass
What Is Molecular Mass?
Molecular mass is the sum of the atomic masses of all atoms in one molecule of a compound. It is expressed in atomic mass units (amu) or grams per mole (g/mol).
Formula for Molecular Mass:
Molecular Mass = Sum of (Number of each atom x Atomic mass of that atom)
Step-by-Step Calculation of Molecular Mass of Urea (CH4N2O)
| Step |
Element |
No. of Atoms |
Atomic Mass (amu) |
Calculation |
Result (amu) |
| 1 |
Carbon (C) |
1 |
12.011 |
1 x 12.011 |
12.011 |
| 2 |
Hydrogen (H) |
4 |
1.008 |
4 x 1.008 |
4.032 |
| 3 |
Nitrogen (N) |
2 |
14.007 |
2 x 14.007 |
28.014 |
| 4 |
Oxygen (O) |
1 |
15.999 |
1 x 15.999 |
15.999 |
| TOTAL |
— |
8 |
— |
Sum of all |
60.056 g/mol |
Molecular Mass of Urea:
60.056 g/mol (commonly rounded to 60.06 g/mol or simply 60 g/mol in board exams)
Molar Mass vs. Molecular Mass
- Molecular Mass: Mass of one molecule of urea = 60.056 amu
- Molar Mass: Mass of one mole (6.022 x 10^23 molecules) of urea = 60.056 g/mol
- For exam purposes: The numerical values are identical; only the units differ (amu vs g/mol)
Complete Formula Table
This section provides every formula related to urea that appears in school and competitive examinations.
A. Identity & Composition Formulas
| Formula Name |
Formula |
Explanation |
Variables |
Exam Use Case |
| Molecular Formula |
CH4N2O |
Exact atomic composition of one urea molecule |
C, H, N, O atoms |
Write the formula of urea |
| Alternative Molecular Formula |
CO(NH2)2 |
Highlights carbonyl + 2 amino groups |
Functional groups |
Show functional groups in urea |
| Expanded Formula |
H2N-CO-NH2 |
Shows connectivity of all atoms |
All atoms and bonds |
Draw structural formula |
| Empirical Formula |
CH4N2O |
Simplest atom ratio (same as molecular) |
Whole number ratio |
Empirical vs molecular formula Qs |
| Molecular Mass Formula |
M = 12+4+28+16 = 60 |
Sum of atomic masses (rounded values) |
Atomic masses of C,H,N,O |
Calculate molecular mass |
B. Mole Concept Formulas Applied to Urea
| Formula Name |
Formula |
Explanation |
Example with Urea |
| Number of Moles |
n = mass (g) / molar mass (g/mol) |
Calculate moles from given mass |
n = 120 / 60 = 2 moles of urea |
| Mass from Moles |
mass = n x M |
Find mass when moles are given |
mass = 3 x 60 = 180 g of urea |
| Number of Molecules |
N = n x 6.022 x 10^23 |
Avogadro’s number application |
1 mol urea = 6.022 x 10^23 molecules |
| Number of Atoms |
Atoms = N x atoms per molecule |
Total atoms in sample |
1 mol urea has 8 x 6.022 x 10^23 atoms |
| Number of N atoms |
N(N) = n x 2 x 6.022 x 10^23 |
Urea has 2 N atoms per molecule |
1 mol urea = 1.204 x 10^24 N atoms |
C. Percentage Composition Formulas
| Element |
Formula for % Composition |
Calculation |
Result |
| % Carbon |
(12/60) x 100 |
12.011/60.056 x 100 |
20.00% |
| % Hydrogen |
(4/60) x 100 |
4.032/60.056 x 100 |
6.71% |
| % Nitrogen |
(28/60) x 100 |
28.014/60.056 x 100 |
46.65% |
| % Oxygen |
(16/60) x 100 |
15.999/60.056 x 100 |
26.64% |
| Total |
— |
Sum must equal 100% |
100.00% |
| Agricultural Significance:
Nitrogen content of urea = 46.65% This is WHY urea is the most popular nitrogen fertilizer. No other solid nitrogen fertilizer provides as high a nitrogen percentage. |
D. Synthesis & Reaction Formulas
| Reaction Name |
Chemical Equation |
Conditions |
Significance |
| Wohler Synthesis (1828) |
NH4CNO –heat–> (NH2)2CO |
Heat |
First lab synthesis of organic from inorganic compound |
| Industrial Synthesis (Bosch-Meiser) |
CO2 + 2NH3 –> (NH2)2CO + H2O |
150-200 atm, 180-200 degC |
Commercial urea production |
| Step 1: Carbamate Formation |
CO2 + 2NH3 –> NH4COONH2 |
High pressure |
Exothermic intermediate step |
| Step 2: Dehydration |
NH4COONH2 –> (NH2)2CO + H2O |
High temp, low pressure |
Endothermic dehydration step |
| Biuret Formation (side reaction) |
2(NH2)2CO –> NH(CONH2)2 + NH3 |
Excess heat |
Undesirable impurity in fertilizer |
| Hydrolysis of Urea |
(NH2)2CO + H2O –> CO2 + 2NH3 |
Urease enzyme or heat |
Biological breakdown in soil |
E. Physical Properties Formulas & Data
| Property |
Formula / Value |
Notes |
| Solubility in Water |
1080 g/L at 20 degC |
Highly soluble; increases with temperature |
| Solubility Product |
Not applicable (highly soluble) |
No Ksp for urea |
| Melting Point |
132.7 degC (405.85 K) |
Decomposes above melting point |
| Density |
1.323 g/cm3 |
At room temperature |
| pKa (conjugate acid) |
~0.18 |
Very weak base |
| Degree of Unsaturation (DoU) |
DoU = (2C + 2 + N – H) / 2 = (2 + 2 + 2 – 4) / 2 = 1 |
1 degree = one C=O double bond |
Urea Synthesis Formula (Wohler & Industrial)
Wohler Synthesis
In 1828, Friedrich Wohler accidentally synthesized urea by heating ammonium cyanate. This event shattered the “vital force” theory which claimed organic compounds could only be produced by living organisms.
Wohler Synthesis Reaction:
NH4CNO –(heat)–> CO(NH2)2Ammonium Cyanate –> UreaKey: Ammonium cyanate (inorganic) converted to urea (organic) a milestone in chemistry history.
Industrial Production — Bosch-Meiser Process
Modern urea is produced industrially from ammonia and carbon dioxide. This is a two-step process:
Step 1 — Carbamate Formation (Exothermic):
CO2 + 2 NH3 –> NH2COONH4 (Ammonium Carbamate)
Conditions: 150-200 atm pressure, temperature around 180-200 degrees C
Step 2 — Dehydration (Endothermic):
NH2COONH4 –> (NH2)2CO + H2O
Conditions: Reduced pressure, high temperature drives water out
Overall Balanced Equation:
CO2 + 2 NH3 –> (NH2)2CO + H2O
| Parameter |
Value / Detail |
| Temperature |
180-200 degrees C |
| Pressure |
150-200 atm |
| Conversion Efficiency |
~70-80% per pass |
| Raw Materials |
Ammonia (NH3) and Carbon Dioxide (CO2) |
| By-product |
Water (H2O) |
| Global Production |
Over 180 million metric tons per year |
Urea-Related Derived Formulas
A. Mole Concept — Formulas with Urea Examples
| Concept |
Formula |
Example with Urea (M = 60 g/mol) |
| Moles from mass |
n = W / M |
n = 180 g / 60 g/mol = 3 mol |
| Mass from moles |
W = n x M |
W = 2.5 x 60 = 150 g |
| Molecules from moles |
N = n x NA |
3 mol x 6.022E23 = 1.807E24 molecules |
| Moles from molecules |
n = N / NA |
1.204E24 / 6.022E23 = 2 mol |
| Mass from molecules |
W = (N / NA) x M |
(1.204E24 / 6.022E23) x 60 = 120 g |
| Atoms of N from moles of urea |
N(N-atoms) = n x 2 x NA |
1 mol x 2 x 6.022E23 = 1.204E24 N atoms |
B. Percentage Composition Formula
General Formula:
% of element = (Mass of element in formula / Molecular mass of compound) x 100For % Nitrogen in Urea:% N = (28 / 60) x 100 = 46.67%
C. Degree of Unsaturation (DBE) Formula
Degree of Unsaturation (DoU) or Double Bond Equivalent (DBE) indicates the number of rings or double bonds in a molecule.
DBE Formula for CnHmNpOq:
DoU = (2C + 2 + N – H) / 2For Urea CH4N2O:DoU = (2(1) + 2 + 2 – 4) / 2DoU = (2 + 2 + 2 – 4) / 2DoU = 2/2 = 1Result: 1 degree of unsaturation = the C=O double bond
D. Fertilizer-Related Formulas
| Formula |
Application |
Urea Example |
| % N = (Total N mass / Formula mass) x 100 |
Calculating nitrogen content of fertilizer |
% N = 28/60 x 100 = 46.67% |
| kg N per bag = (% N / 100) x bag mass (kg) |
Finding actual N in a fertilizer bag |
50 kg bag: 46.67/100 x 50 = 23.33 kg N |
| Fertilizer dose = (Crop N need / % N) x 100 |
Calculating how much fertilizer to apply |
If 50 kg N needed: 50 / 0.4667 = 107 kg urea |
Solved Examples
Question: Find the molecular mass of urea given its formula CH4N2O.
Solution:
- Molecular formula of urea = CH4N2O
- Atomic mass of C = 12, H = 1, N = 14, O = 16 (using rounded values for board exams)
- Molecular mass = 1(12) + 4(1) + 2(14) + 1(16)
- = 12 + 4 + 28 + 16
- = 60 g/mol
Answer:
Molecular mass of urea = 60 g/mol
Question: Calculate the number of moles in 120 g of urea.
Solution:
- Given: mass (W) = 120 g
- Molar mass of urea (M) = 60 g/mol
- Formula: n = W / M
- n = 120 / 60
- n = 2 moles
Answer:
120 g of urea = 2 moles of urea
Question: Find the percentage composition of nitrogen in urea CH4N2O.
Solution:
- Molecular formula = CH4N2O, Molecular mass = 60 g/mol
- Mass of N in one molecule = 2 x 14 = 28 g/mol
- % N = (mass of N / molecular mass) x 100
- % N = (28 / 60) x 100
- % N = 46.67%
Answer:
Nitrogen content in urea = 46.67% (This is why urea is valued as a fertilizer — it provides the highest % N of any solid nitrogenous fertilizer)
Question: Calculate the number of urea molecules in 30 g of urea.
Solution:
- Molar mass of urea = 60 g/mol
- Step 1: Find moles: n = 30 / 60 = 0.5 mol
- Step 2: Number of molecules = n x Avogadro’s number
- N = 0.5 x 6.022 x 10^23
- N = 3.011 x 10^23 molecules
Answer:
30 g of urea contains 3.011 x 10^23 molecules
Question: How many nitrogen atoms are present in 60 g of urea?
Solution:
- Molar mass of urea = 60 g/mol
- Moles of urea = 60/60 = 1 mol
- Each molecule of urea has 2 N atoms
- Moles of N atoms = 1 x 2 = 2 mol
- Number of N atoms = 2 x 6.022 x 10^23 = 1.204 x 10^24 atoms
Answer:
1.204 x 10^24 nitrogen atoms are present in 60 g of urea
Question: How many grams of ammonia (NH3) are needed to produce 60 g of urea? Use: CO2 + 2NH3 –> (NH2)2CO + H2O
Solution:
- Molar mass of urea = 60 g/mol, molar mass of NH3 = 17 g/mol
- Moles of urea to be produced = 60/60 = 1 mol
- From equation: 2 mol NH3 produces 1 mol urea
- Moles of NH3 needed = 2 x 1 = 2 mol
- Mass of NH3 = 2 x 17 = 34 g
Answer:
34 g of ammonia is required to produce 60 g of urea
Common Student Mistakes and How to Avoid Them
| Mistake |
Incorrect |
Correct |
Tip to Remember |
| Wrong atom count for H |
Writing CH2N2O (only 2 H) |
CH4N2O (4 H atoms total — 2 per NH2 group) |
Two NH2 groups = 2 x 2 = 4 H atoms |
| Swapping N and O count |
Writing CH4NO2 |
CH4N2O (2 N, 1 O) |
Nitrogen is 2, Oxygen is 1 — N comes before O in the formula |
| Wrong molecular mass |
Calculating as 56 or 64 |
60 g/mol |
Always: 12 + 4 + 28 + 16 = 60 |
| Confusing urea with uric acid |
Using C5H4N4O3 for urea |
CH4N2O for urea |
Urea is simple (60 g/mol); uric acid is complex (168 g/mol) |
| Wrong IUPAC name |
Calling it carbonate or carbamate |
Carbonamide or diaminomethanone |
The -amide suffix indicates NH2 groups on C=O |
| Using wrong atomic masses |
Using N = 13 or O = 18 |
N = 14, O = 16 |
Memorize: H=1, C=12, N=14, O=16, S=32 |
| Wrong % N calculation |
% N = 14/60 x 100 = 23.3% |
% N = 28/60 x 100 = 46.67% |
There are TWO nitrogen atoms, so mass = 2 x 14 = 28 |
Memory Tricks & Exam Tips
Trick 1: Remember CH4N2O Using a Mnemonic
Mnemonic for CH4N2O:
Carbon Has 4 Neighbors, 2 Nitrogens, 1 Oxygen”C = 1 CarbonH = 4 HydrogensN = 2 NitrogensO = 1 Oxygen
Trick 2: The 60 Rule
The molecular mass of urea is exactly 60 g/mol using rounded atomic masses:
- 12 + 4 + 28 + 16 = 60
- Think: 12-4-28-16 adds up to 60. It is clean and round — easy to remember.
Trick 3: Functional Group Visualization
Picture urea as: Two hands (NH2) shaking around a double-bonded C=O center. That image gives you H2N-CO-NH2 = CO(NH2)2.
Trick 4: The 46% N Fertilizer Rule
Urea = 46% N. This number appears in agriculture questions. Remember: “4-6” as in 46 percent the highest N fertilizer available commercially.
Exam Strategy Tips
- Always write the formula as CH4N2O or CO(NH2)2 — both are accepted in CBSE, ICSE, and competitive exams
- In stoichiometry problems, use molar mass = 60 g/mol unless asked to use precise atomic masses
- In biological/biochemical questions, note that urea is the end product of the urea cycle (ornithine cycle) in mammals
- In agriculture questions, remember that urea provides 46.65% N the highest of any solid nitrogen fertilizer
- When writing the structural formula in organic chemistry, always show the C=O double bond clearly
FAQs About Urea Formula
Q. What is the chemical formula of urea?
The chemical formula of urea is CH4N2O, also written as CO(NH2)2. It contains 1 carbon, 4 hydrogen, 2 nitrogen, and 1 oxygen atom. The IUPAC name is carbonamide. Urea has a molecular mass of 60 g/mol.
Q. What is the molecular formula of urea and its molecular mass?
The molecular formula of urea is CH4N2O. Its molecular mass is 60.06 g/mol (commonly rounded to 60 g/mol). Calculated as: 1(12) + 4(1) + 2(14) + 1(16) = 60 g/mol. This is one of the most commonly asked values in board exams.
Q. What is urea ka formula?
Urea ka formula hai CH4N2O ya CO(NH2)2. Iska molecular mass 60 g/mol hai. Ismein 1 carbon, 4 hydrogen, 2 nitrogen, aur 1 oxygen atom hote hain. IUPAC naam carbonamide hai. Yaha formula CBSE aur state board dono exams ke liye same hai.
Q. What is the IUPAC name of urea?
The IUPAC name of urea is carbonamide. It is also called diaminomethanone in systematic nomenclature. The common name is urea, and it is also known as carbamide in pharmaceutical contexts. All three names refer to the same compound with formula CH4N2O.
Q. What is the structural formula of urea?
The structural formula of urea is H2N-CO-NH2, showing a central carbonyl group (C=O) flanked by two amino groups (NH2). It can also be written as (NH2)2CO or CO(NH2)2. The central carbon is sp2 hybridized, and the molecule has trigonal planar geometry.
Q. How do you calculate the molecular mass of urea?
Use the formula: Molecular mass = sum of (number of atoms x atomic mass) for each element. For urea CH4N2O: C=12, H=4×1=4, N=2×14=28, O=16. Total = 12+4+28+16 = 60 g/mol. Always use rounded atomic masses (H=1, C=12, N=14, O=16) in board exam calculations.
Q. What is the percentage of nitrogen in urea?
The percentage of nitrogen in urea is 46.67%. Calculated as: % N = (mass of N / molecular mass) x 100 = (28/60) x 100 = 46.67%. This is the highest nitrogen content of any solid nitrogen fertilizer, which is why urea is the most widely used agricultural fertilizer worldwide.
Q. Is urea organic or inorganic? Why is this important?
Urea is an organic compound because it contains carbon and is produced by living organisms. However, it was the first organic compound synthesized in a laboratory from inorganic materials (Wohler, 1828). This discovery was historically significant as it disproved the vitalism theory in chemistry.
Learn Urea Formula for Exam Success
Urea is one of chemistry’s most elegant and important compounds. With the formula CH4N2O (or CO(NH2)2), a molecular mass of 60 g/mol, and a nitrogen content of 46.67%, it connects topics across inorganic chemistry, organic chemistry, stoichiometry, agricultural science, and biochemistry.
Let us recap the most critical facts you must remember:
| Fact |
Value / Answer |
| Chemical Formula |
CH4N2O or CO(NH2)2 |
| Alternative Formula |
H2N-CO-NH2 |
| IUPAC Name |
Carbonamide |
| Common Names |
Urea, Carbamide |
| Molecular Mass |
60.06 g/mol (use 60 in exams) |
| % Nitrogen |
46.67% |
| % Carbon |
20.00% |
| % Hydrogen |
6.71% |
| % Oxygen |
26.64% |
| Degree of Unsaturation |
1 (the C=O bond) |
| Hybridization of C |
sp2 |
| Synthesis Reaction |
CO2 + 2NH3 –> (NH2)2CO + H2O |
| Wohler Synthesis |
NH4CNO –heat–> (NH2)2CO |
| Molar Mass for Mole Calculations |
60 g/mol |
| Geometry |
Trigonal planar |
Whether you are preparing for your Class 10 board exams, tackling a Class 12 stoichiometry problem, or studying for NEET and JEE, a firm grasp of the urea formula and its applications will serve you well across multiple chapters and exam formats.
Related Important Formulas List