What is Lactic Acid?
Lactic acid is an organic compound classified as an alpha-hydroxy acid (α-hydroxy acid). It forms naturally in your muscles during intense exercise when oxygen levels are low.
Facts:
- IUPAC Name: 2-hydroxypropanoic acid
- Common Name: Lactic acid
- Type: Carboxylic acid with a hydroxyl group
- Occurrence: Muscle tissue, dairy products, fermented foods
- Function: Energy production, food preservation, cosmetics
Why It Matters:
Understanding lactic acid helps you grasp metabolic processes like anaerobic respiration, fermentation in food science, and industrial applications in pharmaceuticals.
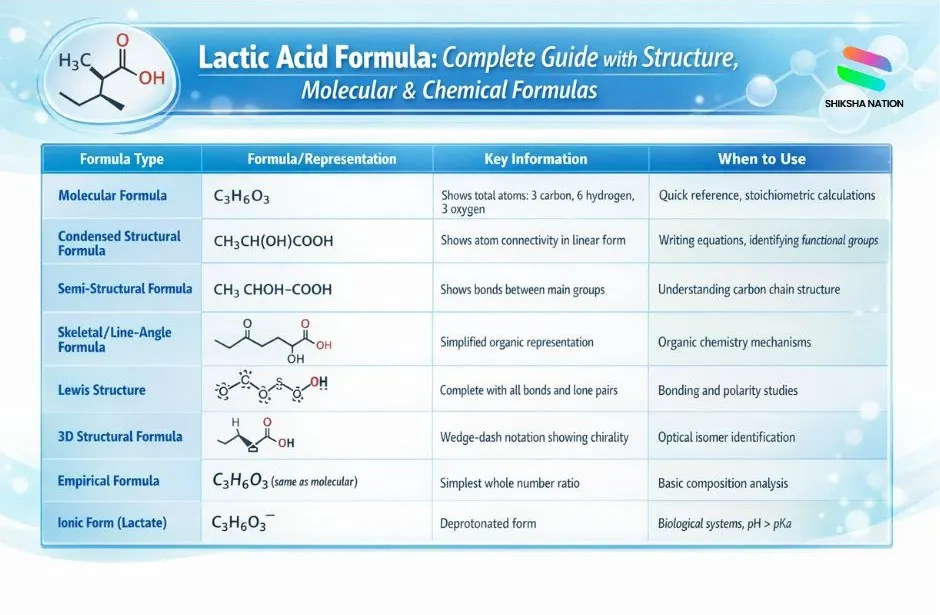
List of Lactic Acid Formulas with Details
| Formula Type |
Formula/Representation |
Key Information |
When to Use |
| Molecular Formula |
C₃H₆O₃ |
Shows total atoms: 3 carbon, 6 hydrogen, 3 oxygen |
Quick reference, stoichiometric calculations |
| Condensed Structural Formula |
CH₃CH(OH)COOH |
Shows atom connectivity in linear form |
Writing equations, identifying functional groups |
| Semi-Structural Formula |
CH₃-CHOH-COOH |
Shows bonds between main groups |
Understanding carbon chain structure |
| Skeletal/Line-Angle Formula |
Zigzag structure with OH groups |
Simplified organic representation |
Organic chemistry mechanisms |
| Lewis Structure |
Complete with all bonds and lone pairs |
Shows electron distribution |
Bonding and polarity studies |
| 3D Structural Formula |
Wedge-dash notation showing chirality |
Displays stereochemistry |
Optical isomer identification |
| Empirical Formula |
C₃H₆O₃ (same as molecular) |
Simplest whole number ratio |
Basic composition analysis |
| Ionic Form (Lactate) |
C₃H₅O₃⁻ |
Deprotonated form |
Biological systems, pH > pKa |
Detailed Formula Explanations
Molecular Formula: C₃H₆O₃
The molecular formula tells you the exact number of each type of atom in one molecule of lactic acid.
Components:
- Carbon (C): 3 atoms
- Hydrogen (H): 6 atoms
- Oxygen (O): 3 atoms
Molecular Weight Calculation:
- C: 3 × 12 = 36 g/mol
- H: 6 × 1 = 6 g/mol
- O: 3 × 16 = 48 g/mol
- Total: 90 g/mol
Example Use: When calculating molar masses for stoichiometry problems or determining percent composition.
Condensed Structural Formula: CH₃CH(OH)COOH
This formula shows how atoms connect without drawing every single bond.
Reading the Structure:
- CH₃: Methyl group (left end)
- CH(OH): Carbon with hydroxyl group attached
- COOH: Carboxylic acid group (right end)
Functional Groups Present:
- Hydroxyl group (-OH): Makes it an alcohol
- Carboxyl group (-COOH): Makes it a carboxylic acid
- Together: α-hydroxy acid (hydroxyl on carbon adjacent to carboxyl)
Example Use: Writing chemical reactions, identifying reaction sites, predicting chemical behavior.
Structural Representations Table
| Representation |
Purpose |
Detail Level |
Academic Level |
| Molecular (C₃H₆O₃) |
Atom count |
Basic |
Grades 9-10 |
| Condensed (CH₃CH(OH)COOH) |
Connectivity |
Intermediate |
Grades 11-12 |
| Expanded/Lewis |
Complete bonding |
Detailed |
College level |
| Skeletal |
Reaction mechanisms |
Advanced |
Organic chemistry |
| 3D Wedge-Dash |
Stereochemistry |
Specialized |
Biochemistry |
Structural Representations
Expanded Structural Formula
In the expanded form, every carbon-hydrogen and carbon-carbon bond is shown explicitly:
H O
| ||
H—C—C—C—O—H
| |
H O
|
H
What It Shows:
- All single bonds between atoms
- Position of each hydrogen atom
- Location of functional groups
- Bond angles (approximately)
Skeletal (Line-Angle) Formula
In organic chemistry, the skeletal formula simplifies representation:
- Carbon atoms at each line intersection and end
- Hydrogen atoms on carbons are implied
- Only heteroatoms (O, N, etc.) and their H atoms are shown
Advantages:
- Cleaner, less cluttered
- Easier to see carbon skeleton
- Standard in advanced organic chemistry
3D Stereochemical Formula
Lactic acid has one chiral center (the carbon bearing the OH group), creating two enantiomers:
L-Lactic Acid (L-(+)-lactic acid):
- Naturally occurring in muscles
- Produced during anaerobic metabolism
- Dextrorotatory (+)
D-Lactic Acid (D-(−)-lactic acid):
- Found in some bacterial fermentation
- Less common in human metabolism
- Levorotatory (−)
Notation:
- Wedge (▲): Bond coming out of the page
- Dash (—): Bond going behind the page
- Solid line: Bond in the plane of the page
Chemical Properties and Variations
Ionization and Lactate Formation
Lactic acid is a weak acid with pKa ≈ 3.86.
Dissociation Equation: CH₃CH(OH)COOH ⇌ CH₃CH(OH)COO⁻ + H⁺
At Different pH Values:
- pH < 3.86: Mostly lactic acid (protonated form)
- pH = 3.86: Equal amounts of acid and lactate
- pH > 3.86: Mostly lactate ion (deprotonated)
- Physiological pH (7.4): Predominantly lactate
Biological Significance: In your body, lactic acid quickly converts to lactate and a hydrogen ion, which is why it’s often called “lactate” in biological contexts.
Related Chemical Forms
| Form |
Formula |
Description |
Context |
| Lactic Acid |
C₃H₆O₃ |
Neutral molecule |
Acidic solutions, pure compound |
| Lactate Ion |
C₃H₅O₃⁻ |
Conjugate base |
Blood, muscle tissue, neutral pH |
| Sodium Lactate |
C₃H₅NaO₃ |
Salt form |
Food preservative, medical IV fluids |
| Calcium Lactate |
C₆H₁₀CaO₆ |
Calcium salt |
Dietary supplement |
| Polylactic Acid (PLA) |
(C₃H₄O₂)ₙ |
Polymer |
Biodegradable plastics |
Solved Examples
Example 1: Calculating Percentage Composition
Question: Calculate the percentage composition of each element in lactic acid (C₃H₆O₃).
Solution: Molecular mass = 90 g/mol
Carbon: (3 × 12) / 90 × 100 = 36/90 × 100 = 40%
Hydrogen: (6 × 1) / 90 × 100 = 6/90 × 100 = 6.67%
Oxygen: (3 × 16) / 90 × 100 = 48/90 × 100 = 53.33%
Answer: C: 40%, H: 6.67%, O: 53.33%
Example 2: Identifying Functional Groups
Question: Identify all functional groups in the lactic acid structure CH₃CH(OH)COOH.
Solution:
Functional groups present:
- Carboxyl group (-COOH): Present at the right end
- Makes it a carboxylic acid
- Acidic hydrogen
- Can form esters and amides
- Hydroxyl group (-OH): Attached to the middle carbon
- Makes it an alcohol
- Secondary alcohol (attached to carbon with two other carbons)
- Can undergo oxidation
Classification: Alpha-hydroxy acid (α-hydroxy acid) because the -OH is on the carbon adjacent (alpha position) to the -COOH group.
Answer: Carboxyl group and hydroxyl group; classified as an α-hydroxy carboxylic acid.
Example 3: Stoichiometry Problem
Question: How many moles of lactic acid are present in 450 g of pure lactic acid?
Solution:
Given:
- Mass of lactic acid = 450 g
- Molar mass of C₃H₆O₃ = 90 g/mol
Formula: Number of moles = Mass / Molar mass
Calculation: Number of moles = 450 g / 90 g/mol = 5 moles
Answer: 5 moles of lactic acid
Example 4: Writing Neutralization Reaction
Question: Write the balanced equation for the neutralization of lactic acid with sodium hydroxide.
Solution:
Reactants:
- Lactic acid: CH₃CH(OH)COOH
- Sodium hydroxide: NaOH
Products:
- Sodium lactate: CH₃CH(OH)COONa
- Water: H₂O
Balanced Equation: CH₃CH(OH)COOH + NaOH → CH₃CH(OH)COONa + H₂O
Answer: The balanced equation shows a 1:1 molar ratio, producing sodium lactate and water.
Example 5: Determining Empirical Formula
Question: A compound contains 40% C, 6.67% H, and 53.33% O by mass. Show that its empirical formula is C₃H₆O₃.
Solution:
Step 1: Assume 100 g sample
- C: 40 g
- H: 6.67 g
- O: 53.33 g
Step 2: Convert to moles
- C: 40/12 = 3.33 mol
- H: 6.67/1 = 6.67 mol
- O: 53.33/16 = 3.33 mol
Step 3: Divide by smallest (3.33)
- C: 3.33/3.33 = 1
- H: 6.67/3.33 = 2
- O: 3.33/3.33 = 1
Step 4: Multiply to get whole numbers (×3)
- C: 1 × 3 = 3
- H: 2 × 3 = 6
- O: 1 × 3 = 3
Answer: Empirical formula = C₃H₆O₃ (which is also the molecular formula for lactic acid)
Common Mistakes Students Make
Mistake 1: Confusing Lactic Acid with Lactose
Wrong Assumption: Thinking lactic acid is the sugar in milk.
Correction: Lactose is milk sugar (C₁₂H₂₂O₁₁). Lactic acid (C₃H₆O₃) is produced when bacteria ferment lactose, but they’re different compounds.
Mistake 2: Writing Incorrect Structural Formula
Common Error: CH₂OHCH₂COOH (wrong position of OH)
Correct Formula: CH₃CH(OH)COOH (OH on the middle carbon, not the end)
Why It Matters: The position of the hydroxyl group determines the compound’s properties and makes it an α-hydroxy acid.
Mistake 3: Forgetting About Stereoisomers
Incomplete Answer: Only mentioning one form of lactic acid.
Complete Answer: Lactic acid exists as two enantiomers (L and D forms) due to the chiral center at C-2.
Exam Tip: If asked about optical activity or isomers, always mention both L and D forms.
Mistake 4: Incorrect Molecular Mass Calculation
Common Error: Calculating as 88 g/mol or 92 g/mol
Correct Calculation:
- C₃: 3 × 12 = 36
- H₆: 6 × 1 = 6
- O₃: 3 × 16 = 48
- Total: 90 g/mol
Mistake 5: Confusing Lactate and Lactic Acid
Error: Using the terms interchangeably without context.
Clarification:
- Lactic acid (C₃H₆O₃): The protonated form
- Lactate (C₃H₅O₃⁻): The deprotonated ion
- At body pH, mostly exists as lactate
Memory Tricks and Tips
Trick 1: “3-6-3 Rule”
Remember: C₃H₆O₃ → “Three-Six-Three”
Think: “Three carbons, six hydrogens, three oxygens”
Trick 2: Functional Group Memory
“COOH on the end, OH in the middle”
This reminds you that:
- Carboxylic acid group (-COOH) is at one end
- Hydroxyl group (-OH) is on the middle carbon
- Methyl group (CH₃) is at the other end
Trick 3: Alpha Position Reminder
“Alpha means Adjacent”
The α-hydroxy means the -OH is on the carbon adjacent (next to) the -COOH group.
Trick 4: Molecular Mass Memory
“Lactic acid weighs 90”
Simple phrase to remember the molar mass: 90 g/mol
Trick 5: L vs D Forms
“L for Life”
L-lactic acid is the form produced in living organisms (muscles, tissues). D-form is less common in human biology.
Conclusion
Mastering the lactic acid formula goes beyond memorizing C₃H₆O₃. Understanding its structural variations, from the condensed formula CH₃CH(OH)COOH to the three-dimensional stereochemical representation, equips you with the knowledge to tackle diverse chemistry problems.
Remember these key points: the molecular formula contains 3 carbons, 6 hydrogens, and 3 oxygens with a molar mass of 90 g/mol. The structure features both a carboxyl group and a hydroxyl group, making it an α-hydroxy acid. It exists in two enantiomeric forms, with L-lactic acid being the biologically significant form in human metabolism.
Whether you’re solving stoichiometry problems, identifying functional groups, or explaining metabolic pathways, this comprehensive understanding of lactic acid formulas will serve you well in exams and practical applications. Keep practicing with the solved examples, avoid the common mistakes outlined here, and use the memory tricks to reinforce your learning.
Frequently Asked Questions about Lactic Acid
Q. What is the chemical formula of lactic acid?
The chemical formula of lactic acid is C₃H₆O₃. This molecular formula indicates that each molecule contains three carbon atoms, six hydrogen atoms, and three oxygen atoms, with a molar mass of 90 g/mol.
Q. How do you write the structural formula of lactic acid?
The structural formula is written as CH₃CH(OH)COOH. This shows a methyl group (CH₃), a carbon with a hydroxyl group CH(OH), and a carboxylic acid group (COOH) connected in sequence from left to right.
Q. Is lactic acid the same as lactate?
No, they’re different. Lactic acid (C₃H₆O₃) is the protonated acidic form. Lactate (C₃H₅O₃⁻) is the conjugate base formed when lactic acid loses a proton. In blood and muscles at physiological pH, it exists predominantly as lactate.
Q. What functional groups are present in lactic acid?
Lactic acid contains two functional groups: a carboxyl group (-COOH) making it a carboxylic acid, and a hydroxyl group (-OH) making it also an alcohol. This combination classifies it as an alpha-hydroxy acid or α-hydroxy carboxylic acid.
Q. What is the molecular weight of lactic acid?
The molecular weight of lactic acid is 90 g/mol, calculated from its molecular formula C₃H₆O₃: (3×12) + (6×1) + (3×16) = 36 + 6 + 48 = 90 g/mol. This value is essential for stoichiometric calculations.
Q. Does lactic acid have stereoisomers?
Yes, lactic acid has one chiral center and exists as two enantiomers: L-(+)-lactic acid (naturally produced in human muscles) and D-(−)-lactic acid (found in some bacterial fermentation). They are mirror images of each other with different optical rotations.
Q. What is the empirical formula of lactic acid?
The empirical formula of lactic acid is C₃H₆O₃, which happens to be the same as its molecular formula. This represents the simplest whole number ratio of atoms present: 3 carbons, 6 hydrogens, and 3 oxygens.
Q. How is lactic acid formed in muscles?
During intense exercise when oxygen is limited, muscles break down glucose through anaerobic glycolysis, converting pyruvate to lactic acid. The reaction is catalyzed by lactate dehydrogenase enzyme: pyruvate + NADH + H⁺ → lactate + NAD⁺, helping regenerate NAD⁺ for continued glycolysis.