What is Citric Acid?
Citric acid is a weak organic acid found naturally in citrus fruits like lemons, oranges, and limes. It gives these fruits their characteristic sour taste.
Key Properties:
- Weak tricarboxylic acid (has three carboxyl groups)
- White crystalline powder at room temperature
- Highly soluble in water
- Used as a preservative, flavoring agent, and cleaning agent
Why it matters in chemistry: Citric acid is a perfect example of organic acids, functional groups, and molecular structure. Understanding its various formulas helps you grasp fundamental chemistry concepts.
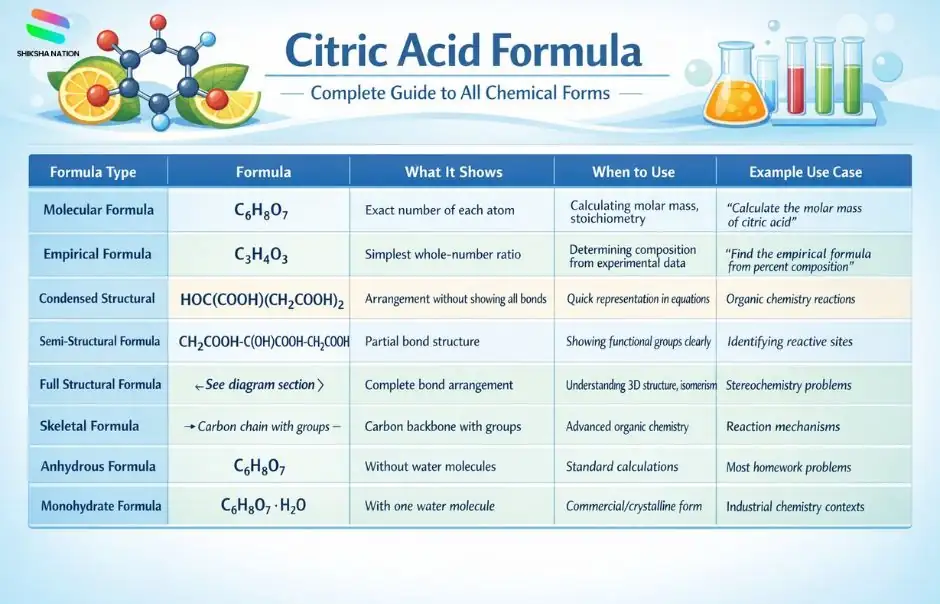
Complete List of Citric Acid Formulas
Here’s every formula variation you need to know about citric acid, organized in a comprehensive table:
| Formula Type |
Formula |
What It Shows |
When to Use |
Example Use Case |
| Molecular Formula |
C₆H₈O₇ |
Exact number of each atom |
Calculating molar mass, stoichiometry |
“Calculate the molar mass of citric acid” |
| Empirical Formula |
C₆H₈O₇ |
Simplest whole-number ratio |
Determining composition from experimental data |
“Find the empirical formula from percent composition” |
| Condensed Structural Formula |
HOC(COOH)(CH₂COOH)₂ |
Arrangement without showing all bonds |
Quick representation in equations |
Organic chemistry reactions |
| Semi-Structural Formula |
CH₂COOH-C(OH)COOH-CH₂COOH |
Partial bond structure |
Showing functional groups clearly |
Identifying reactive sites |
| Full Structural Formula |
[See diagram section] |
Complete bond arrangement |
Understanding 3D structure, isomerism |
Stereochemistry problems |
| Skeletal Formula |
[Carbon chain with groups] |
Carbon backbone with groups |
Advanced organic chemistry |
Reaction mechanisms |
| Anhydrous Formula |
C₆H₈O₇ |
Without water molecules |
Standard calculations |
Most homework problems |
| Monohydrate Formula |
C₆H₈O₇·H₂O |
With one water molecule |
Commercial/crystalline form |
Industrial chemistry contexts |
Understanding Each Formula Type
Molecular Formula: C₆H₈O₇
What it is: The molecular formula shows the exact number of each type of atom in one citric acid molecule.
Breaking it down:
- 6 Carbon atoms (C₆)
- 8 Hydrogen atoms (H₈)
- 7 Oxygen atoms (O₇)
Molar Mass Calculation:
- C: 6 × 12.01 = 72.06 g/mol
- H: 8 × 1.008 = 8.064 g/mol
- O: 7 × 16.00 = 112.00 g/mol
- Total: 192.124 g/mol
Empirical Formula: C₆H₈O₇
- Special note: For citric acid, the empirical and molecular formulas are identical because the ratio 6:8:7 is already in its simplest whole-number form.
- What it tells you: The simplest ratio of atoms present. If you were to determine citric acid’s composition from combustion analysis, you’d arrive at this ratio.
Structural Formula
The structural formula shows how atoms are bonded together. Citric acid has:
Three carboxyl groups (-COOH): These make it an acid One hydroxyl group (-OH): Attached to the central carbon Two methylene groups (-CH₂-): Connecting the structure
Condensed form: HOC(COOH)(CH₂COOH)₂
This notation shows:
- A central carbon with OH and COOH
- Two CH₂COOH branches
Anhydrous vs. Monohydrate
Anhydrous citric acid: C₆H₈O₇ (no water)
Citric acid monohydrate: C₆H₈O₇·H₂O
The monohydrate form includes one water molecule per citric acid molecule. This is the common commercial form.
Molar mass of monohydrate: 192.124 + 18.015 = 210.139 g/mol
Citric Acid Formula Structure Explained
Functional Groups Present
- Three carboxylic acid groups (-COOH)
- Make citric acid a triprotic acid
- Can donate three hydrogen ions (H⁺)
- Responsible for sour taste and acidity
- One hydroxyl group (-OH)
- Attached to the central carbon
- Increases water solubility
- Can participate in esterification
- Carbon backbone
- Six-carbon chain structure
- Central carbon is tertiary (bonded to three other carbons and one OH)
IUPAC Name
2-hydroxypropane-1,2,3-tricarboxylic acid
This systematic name tells you:
- “2-hydroxy” = OH group on carbon 2
- “propane” = three-carbon backbone
- “1,2,3-tricarboxylic acid” = COOH groups on carbons 1, 2, and 3
Solved Examples
Example 1: Calculate Molar Mass
Question: What is the molar mass of anhydrous citric acid?
Solution: Formula: C₆H₈O₇
Step 1: Multiply atomic masses
- Carbon: 6 × 12.01 = 72.06
- Hydrogen: 8 × 1.008 = 8.064
- Oxygen: 7 × 16.00 = 112.00
Step 2: Add them together 72.06 + 8.064 + 112.00 = 192.124 g/mol
Example 2: Finding Number of Moles
Question: How many moles are in 96.062 g of citric acid?
Solution: Number of moles = mass ÷ molar mass = 96.062 g ÷ 192.124 g/mol = 0.5 moles
Example 3: Percent Composition
Question: What is the percent composition of carbon in citric acid?
Solution: Step 1: Mass of carbon = 6 × 12.01 = 72.06 g Step 2: Total molar mass = 192.124 g/mol Step 3: Percent = (72.06 ÷ 192.124) × 100 = 37.51% carbon
Example 4: Converting Monohydrate to Anhydrous
Question: If you have 10.0 g of citric acid monohydrate, how much anhydrous citric acid does it contain?
Solution: Molar mass of monohydrate = 210.139 g/mol Molar mass of anhydrous = 192.124 g/mol
Moles of monohydrate = 10.0 ÷ 210.139 = 0.0476 mol Mass of anhydrous = 0.0476 × 192.124 = 9.14 g
Common Mistakes Students Make
Mistake 1: Confusing Molecular and Empirical Formulas
Wrong thinking: “The empirical formula must be simpler than C₆H₈O₇”
Reality: For citric acid, they’re the same. Not all compounds have different empirical and molecular formulas.
Fix: Always reduce to simplest ratio first, then check if it matches the molecular formula.
Mistake 2: Forgetting the Hydroxyl Group
Wrong structure: Writing only three COOH groups without the central -OH
Reality: Citric acid has BOTH three carboxyl groups AND one hydroxyl group.
Fix: Remember “hydroxy” in the IUPAC name indicates the -OH group.
Mistake 3: Wrong Molar Mass Calculation
Common error: Using 210 g/mol for all problems
Reality: Standard anhydrous form is 192.124 g/mol; monohydrate is 210.139 g/mol.
Fix: Check whether the problem specifies anhydrous or monohydrate.
Mistake 4: Incorrect Structural Drawing
Common error: Drawing citric acid as a straight chain
Reality: The central carbon is tertiary with branches.
Fix: Start with the central carbon, add OH and COOH to it, then add the two CH₂COOH branches.
Memory Tricks & Exam Tips
Mnemonic for the Formula
“6 Carbons, 8 Hydrogens, 7 Oxygens = C₆H₈O₇”
Think: “Citrus Has Organic acids” → Remember the order C-H-O, then 6-8-7
Quick Recognition
The numbers 6-8-7 decrease by 1 then increase by 1: 6, (6+2), (6+1). Not perfect, but close enough for memory!
Formula Writing Tips
- Start with carbon: Write C₆ first
- Add hydrogen: H₈ (slightly more than carbon)
- Finish with oxygen: O₇ (highest number)
Exam Strategy
When asked “What is the citric acid formula?” without specification:
- Default to molecular formula: C₆H₈O₇
- This is the most commonly requested form
When structural formula is needed:
- Read carefully: “structural,” “structure,” or “arrangement” are key words
- Provide the condensed form if short on time: HOC(COOH)(CH₂COOH)₂
Calculation Shortcuts
Molar mass quick check: Rough estimate: 6(12) + 8(1) + 7(16) = 72 + 8 + 112 = 192 g/mol
This matches the precise value, so you can verify your calculations quickly.
Conclusion
Understanding citric acid formulas isn’t about memorizing random numbers it’s about recognizing patterns and knowing when to use each form. Whether you’re calculating molar mass with C₆H₈O₇, drawing structures with HOC(COOH)(CH₂COOH)₂, or converting between anhydrous and monohydrate forms, each formula serves a specific purpose in chemistry.
The molecular formula C₆H₈O₇ is your go-to for calculations and stoichiometry. The structural formulas help you understand how citric acid behaves in reactions. And knowing the difference between anhydrous and monohydrate forms prevents costly calculation errors.
FAQs about Citric Acid Formula
Q. What is the chemical formula of citric acid?
The chemical formula of citric acid is C₆H₈O₇. This molecular formula indicates citric acid contains 6 carbon atoms, 8 hydrogen atoms, and 7 oxygen atoms per molecule, with a molar mass of 192.124 g/mol.
Q. What is the difference between citric acid molecular formula and empirical formula?
For citric acid, both the molecular and empirical formulas are C₆H₈O₇. Unlike many compounds, citric acid’s atom ratio (6:8:7) is already in its simplest whole-number form, making the empirical and molecular formulas identical.
Q. What is citric acid monohydrate formula?
Citric acid monohydrate has the formula C₆H₈O₇·H₂O. This is the common commercial form where each citric acid molecule is associated with one water molecule, giving it a molar mass of 210.139 g/mol instead of 192.124 g/mol.
Q. How do you write the structural formula of citric acid?
The condensed structural formula is HOC(COOH)(CH₂COOH)₂. This shows a central carbon with a hydroxyl group (OH) and carboxyl group (COOH), plus two CH₂COOH branches, representing citric acid’s three acid groups and one alcohol group.
Q. Why does citric acid have three COOH groups?
Citric acid is a tricarboxylic acid, meaning it has three carboxylic acid groups (-COOH). These three groups make it capable of donating three hydrogen ions (triprotic acid) and are responsible for its acidic properties and sour taste.
Q. Is C₆H₈O₇ the same for both natural and synthetic citric acid?
Yes, both natural citric acid (from citrus fruits) and synthetic citric acid (from fermentation) have the identical molecular formula C₆H₈O₇. The chemical structure is the same regardless of the source, though production methods differ.
Q. What is the molar mass of citric acid?
The molar mass of anhydrous citric acid (C₆H₈O₇) is 192.124 g/mol. For citric acid monohydrate (C₆H₈O₇·H₂O), the molar mass is 210.139 g/mol. Always check which form your problem requires before calculating.
Q. How many oxygen atoms are in citric acid formula?
Citric acid (C₆H₈O₇) contains 7 oxygen atoms per molecule. Six oxygen atoms are in the three carboxyl groups (-COOH), and one oxygen atom is in the hydroxyl group (-OH) attached to the central carbon.